Ribosome-associated complex binds to ribosomes in close proximity of Rpl31 at the exit of the polypeptide tunnel in yeast
- PMID: 18829863
- PMCID: PMC2592665
- DOI: 10.1091/mbc.e08-06-0661
Ribosome-associated complex binds to ribosomes in close proximity of Rpl31 at the exit of the polypeptide tunnel in yeast
Abstract
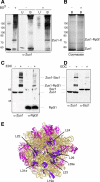
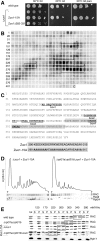
Ribosome-associated complex (RAC) consists of the Hsp40 homolog Zuo1 and the Hsp70 homolog Ssz1. The chaperone participates in the biogenesis of newly synthesized polypeptides. Here we have identified yeast Rpl31, a component of the large ribosomal subunit, as a contact point of RAC at the polypeptide tunnel exit. Rpl31 is encoded by RPL31a and RPL31b, two closely related genes. Delta rpl31a Delta rpl31b displayed slow growth and sensitivity to low as well as high temperatures. In addition, Delta rpl31a Delta rpl31b was highly sensitive toward aminoglycoside antibiotics and suffered from defects in translational fidelity. With the exception of sensitivity at elevated temperature, the phenotype resembled yeast strains lacking one of the RAC subunits or Rpl39, another protein localized at the tunnel exit. Defects of Delta rpl31a Delta rpl31b Delta zuo1 did not exceed that of Delta rpl31a Delta rpl31b or Delta zuo1. However, the combined deletion of RPL31a, RPL31b, and RPL39 was lethal. Moreover, RPL39 was a multicopy suppressor, whereas overexpression of RAC failed to rescue growth defects of Delta rpl31a Delta rpl31b. The findings are consistent with a model in that Rpl31 and Rpl39 independently affect a common ribosome function, whereas Rpl31 and RAC are functionally interdependent. Rpl31, while not essential for binding of RAC to the ribosome, might be involved in proper function of the chaperone complex.
Figures


Similar articles
-
Functional characterization of the atypical Hsp70 subunit of yeast ribosome-associated complex.J Biol Chem. 2007 Nov 23;282(47):33977-84. doi: 10.1074/jbc.M706737200. Epub 2007 Sep 27. J Biol Chem. 2007. PMID: 17901048
-
Structural remodeling of ribosome associated Hsp40-Hsp70 chaperones during co-translational folding.Nat Commun. 2022 Jun 14;13(1):3410. doi: 10.1038/s41467-022-31127-4. Nat Commun. 2022. PMID: 35701497 Free PMC article.
-
Structural characterization of a eukaryotic chaperone--the ribosome-associated complex.Nat Struct Mol Biol. 2013 Jan;20(1):23-8. doi: 10.1038/nsmb.2447. Epub 2012 Dec 2. Nat Struct Mol Biol. 2013. PMID: 23202586
-
Two chaperones locked in an embrace: structure and function of the ribosome-associated complex RAC.Nat Struct Mol Biol. 2017 Aug 3;24(8):611-619. doi: 10.1038/nsmb.3435. Nat Struct Mol Biol. 2017. PMID: 28771464 Review.
-
Dual role of ribosome-associated chaperones in prion formation and propagation.Curr Genet. 2016 Nov;62(4):677-685. doi: 10.1007/s00294-016-0586-2. Epub 2016 Mar 11. Curr Genet. 2016. PMID: 26968706 Review.
Cited by
-
Dual interaction of the Hsp70 J-protein cochaperone Zuotin with the 40S and 60S ribosomal subunits.Nat Struct Mol Biol. 2016 Nov;23(11):1003-1010. doi: 10.1038/nsmb.3299. Epub 2016 Sep 26. Nat Struct Mol Biol. 2016. PMID: 27669034 Free PMC article.
-
Dual binding mode of the nascent polypeptide-associated complex reveals a novel universal adapter site on the ribosome.J Biol Chem. 2010 Jun 18;285(25):19679-87. doi: 10.1074/jbc.M109.092536. Epub 2010 Apr 21. J Biol Chem. 2010. PMID: 20410297 Free PMC article.
-
Structure and evolution of the 4-helix bundle domain of Zuotin, a J-domain protein co-chaperone of Hsp70.PLoS One. 2019 May 15;14(5):e0217098. doi: 10.1371/journal.pone.0217098. eCollection 2019. PLoS One. 2019. PMID: 31091298 Free PMC article.
-
Mechanistic insight into eukaryotic 60S ribosomal subunit biogenesis by cryo-electron microscopy.RNA. 2016 Nov;22(11):1643-1662. doi: 10.1261/rna.057927.116. RNA. 2016. PMID: 27875256 Free PMC article. Review.
-
Interaction of the cotranslational Hsp70 Ssb with ribosomal proteins and rRNA depends on its lid domain.Nat Commun. 2016 Nov 24;7:13563. doi: 10.1038/ncomms13563. Nat Commun. 2016. PMID: 27882919 Free PMC article.
References
-
- Aguilera J., Randez-Gil F., Prieto J. A. Cold response in Saccharomyces cerevisiae: new functions for old mechanisms. FEMS Microbiol. Rev. 2007;31:327–341. - PubMed
-
- Ban N., Nissen P., Hansen J., Moore P. B., Steitz T. A. The complete atomic structure of the large ribosomal subunit at 2.4 A resolution. Science. 2000;289:905–920. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

