BCL11B enhances TCR/CD28-triggered NF-kappaB activation through up-regulation of Cot kinase gene expression in T-lymphocytes
- PMID: 18831712
- PMCID: PMC2639648
- DOI: 10.1042/BJ20080925
BCL11B enhances TCR/CD28-triggered NF-kappaB activation through up-regulation of Cot kinase gene expression in T-lymphocytes
Abstract
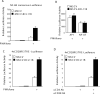
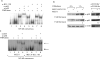
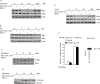
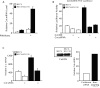
BCL11B is a transcriptional regulator with an important role in T-cell development and leukaemogenesis. We demonstrated recently that BCL11B controls expression from the IL (interleukin)-2 promoter through direct binding to the US1 (upstream site 1). In the present study, we provide evidence that BCL11B also participates in the activation of IL-2 gene expression by enhancing NF-kappaB (nuclear factor kappaB) activity in the context of TCR (T-cell receptor)/CD28-triggered T-cell activation. Enhanced NF-kappaB activation is not a consequence of BCL11B binding to the NF-kappaB response elements or association with the NF-kappaB-DNA complexes, but rather the result of higher translocation of NF-kappaB to the nucleus caused by enhanced degradation of IkappaB (inhibitor of NF-kappaB). The enhanced IkappaB degradation in cells with increased levels of BCL11B was specific for T-cells activated through the TCR, but not for cells activated through TNFalpha (tumour necrosis factor alpha) or UV light, and was caused by increased activity of IkappaB kinase, as indicated by its increase in phosphorylation. As BCL11B is a transcription factor, we investigated whether the expression of genes upstream of IkappaB kinase in the TCR/CD28 signalling pathway was affected by increased BCL11B expression, and found that Cot (cancer Osaka thyroid oncogene) kinase mRNA levels were elevated. Cot kinase is known to promote enhanced IkappaB kinase activity, which results in the phosphorylation and degradation of IkappaB and activation of NF-kappaB. The implied involvement of Cot kinase in BCL11B-mediated NF-kappaB activation in response to TCR activation is supported by the fact that a Cot kinase dominant-negative mutant or Cot kinase siRNA (small interfering RNA) knockdown blocked BCL11B-mediated NF-kappaB activation. In support of our observations, in the present study we report that BCL11B enhances the expression of several other NF-kappaB target genes, in addition to IL-2. In addition, we provide evidence that BCL11B associates with intron 2 of the Cot kinase gene to regulate its expression.
Figures



Similar articles
-
Mechanism responsible for T-cell antigen receptor- and CD28- or interleukin 1 (IL-1) receptor-initiated regulation of IL-2 gene expression by NF-kappaB.Mol Cell Biol. 1998 Jun;18(6):3140-8. doi: 10.1128/MCB.18.6.3140. Mol Cell Biol. 1998. PMID: 9584155 Free PMC article.
-
Vav cooperates with CD28 to induce NF-kappaB activation via a pathway involving Rac-1 and mitogen-activated kinase kinase 1.Eur J Immunol. 2002 Feb;32(2):447-56. doi: 10.1002/1521-4141(200202)32:2<447::AID-IMMU447>3.0.CO;2-5. Eur J Immunol. 2002. PMID: 11813163
-
ZAP-70 and SLP-76 regulate protein kinase C-theta and NF-kappa B activation in response to engagement of CD3 and CD28.J Immunol. 2001 May 1;166(9):5654-64. doi: 10.4049/jimmunol.166.9.5654. J Immunol. 2001. PMID: 11313406
-
Regulation and function of IKK and IKK-related kinases.Sci STKE. 2006 Oct 17;2006(357):re13. doi: 10.1126/stke.3572006re13. Sci STKE. 2006. PMID: 17047224 Review.
-
Regulation of NF-κB induction by TCR/CD28.Immunol Res. 2011 Aug;50(2-3):113-7. doi: 10.1007/s12026-011-8216-z. Immunol Res. 2011. PMID: 21717079 Free PMC article. Review.
Cited by
-
Critical role of Bcl11b in suppressor function of T regulatory cells and prevention of inflammatory bowel disease.J Exp Med. 2011 Sep 26;208(10):2069-81. doi: 10.1084/jem.20102683. Epub 2011 Aug 29. J Exp Med. 2011. PMID: 21875956 Free PMC article.
-
Bcl11 Transcription Factors Regulate Cortical Development and Function.Front Mol Neurosci. 2020 Apr 8;13:51. doi: 10.3389/fnmol.2020.00051. eCollection 2020. Front Mol Neurosci. 2020. PMID: 32322190 Free PMC article. Review.
-
Using mouse models to study function of transcriptional factors in T cell development.Cell Regen. 2012 Oct 10;1(1):8. doi: 10.1186/2045-9769-1-8. eCollection 2012. Cell Regen. 2012. PMID: 25408871 Free PMC article. Review.
-
Construction of a ceRNA-based lncRNA-mRNA network to identify functional lncRNAs in polycystic ovarian syndrome.Aging (Albany NY). 2021 Mar 10;13(6):8481-8496. doi: 10.18632/aging.202659. Epub 2021 Mar 10. Aging (Albany NY). 2021. PMID: 33714202 Free PMC article.
-
Multilayered specification of the T-cell lineage fate.Immunol Rev. 2010 Nov;238(1):150-68. doi: 10.1111/j.1600-065X.2010.00964.x. Immunol Rev. 2010. PMID: 20969591 Free PMC article. Review.
References
-
- Wakabayashi Y, Watanabe H, Inoue J, Takeda N, Sakata J, Mishima Y, Hitomi J, Yamamoto T, Utsuyama M, Niwa O, Aizawa S, Kominami R. Bcl11b is required for differentiation and survival of alphabeta T lymphocytes. Nat Immunol. 2003;4:533–539. - PubMed
-
- Avram D, Fields A, Pretty On Top K, Nevrivy DJ, Ishmael JE, Leid M. Isolation of a novel family of C(2)H(2) zinc finger proteins implicated in transcriptional repression mediated by chicken ovalbumin upstream promoter transcription factor (COUP-TF) orphan nuclear receptors. J Biol Chem. 2000;275:10315–10322. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

