Autonomous initiation and propagation of action potentials in neurons of the subthalamic nucleus
- PMID: 18832425
- PMCID: PMC2655394
- DOI: 10.1113/jphysiol.2008.155861
Autonomous initiation and propagation of action potentials in neurons of the subthalamic nucleus
Erratum in
- J Physiol. 2009 Feb 1;587(Pt 3):709
Abstract
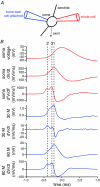
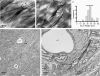
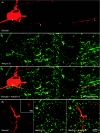
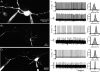
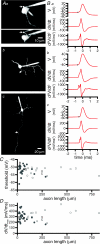
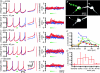
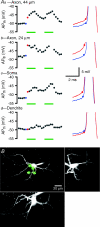
The activity of the subthalamic nucleus (STN) is intimately related to movement and is generated, in part, by voltage-dependent Na(+) (Na(v)) channels that drive autonomous firing. In order to determine the principles underlying the initiation and propagation of action potentials in STN neurons, 2-photon laser scanning microscopy was used to guide tight-seal whole-cell somatic and loose-seal cell-attached axonal/dendritic patch-clamp recordings and compartment-selective ion channel manipulation in rat brain slices. Action potentials were first detected in a region that corresponded most closely to the unmyelinated axon initial segment, as defined by Golgi and ankyrin G labelling. Following initiation, action potentials propagated reliably into axonal and somatodendritic compartments with conduction velocities of approximately 5 m s(-1) and approximately 0.7 m s(-1), respectively. Action potentials generated by neurons with axons truncated within or beyond the axon initial segment were not significantly different. However, axon initial segment and somatic but not dendritic or more distal axonal application of low [Na(+)] ACSF or the selective Na(v) channel blocker tetrodotoxin consistently depolarized action potential threshold. Finally, somatodendritic but not axonal application of GABA evoked large, rapid inhibitory currents in concordance with electron microscopic analyses, which revealed that the somatodendritic compartment was the principal target of putative inhibitory inputs. Together the data are consistent with the conclusions that in STN neurons the axon initial segment and soma express an excess of Na(v) channels for the generation of autonomous activity, while synaptic activation of somatodendritic GABA(A) receptors regulates the axonal initiation of action potentials.
Figures




Comment in
-
Subthalamic firing without an end, but now with a beginning.J Physiol. 2008 Dec 1;586(23):5603. doi: 10.1113/jphysiol.2008.164806. J Physiol. 2008. PMID: 19043119 Free PMC article. No abstract available.
References
-
- Afsharpour S. Light microscopic analysis of Golgi-impregnated rat subthalamic neurons. J Comp Neurol. 1985;236:1–13. - PubMed
-
- Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–375. - PubMed
-
- Alle H, Geiger JR. Combined analog and action potential coding in hippocampal mossy fibers. Science. 2006;311:1290–1293. - PubMed
-
- Alvarez L, Macias R, Lopez G, Alvarez E, Pavon N, Rodriguez-Oroz MC, Juncos JL, Maragoto C, Guridi J, Litvan I, Tolosa ES, Koller W, Vitek J, DeLong MR, Obeso JA. Bilateral subthalamotomy in Parkinson's disease: initial and long-term response. Brain. 2005;128:570–583. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

