Bending flexibility of actin filaments during motor-induced sliding
- PMID: 18835897
- PMCID: PMC2599860
- DOI: 10.1529/biophysj.108.140335
Bending flexibility of actin filaments during motor-induced sliding
Abstract
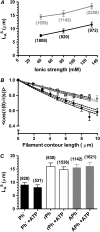
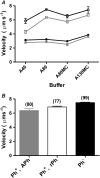
Muscle contraction and other forms of cell motility occur as a result of cyclic interactions between myosin molecules and actin filaments. Force generation is generally attributed to ATP-driven structural changes in myosin, whereas a passive role is ascribed to actin. However, some results challenge this view, predicting structural changes in actin during motor activity, e.g., when the actin filaments slide on a myosin-coated surface in vitro. Here, we analyzed statistical properties of the sliding filament paths, allowing us to detect changes of this type. It is interesting to note that evidence for substantial structural changes that led to increased bending flexibility of the filaments was found in phalloidin-stabilized, but not in phalloidin-free, actin filaments. The results are in accordance with the idea that a high-flexibility structural state of actin is a prerequisite for force production, but not the idea that a low-to-high flexibility transition of the actin filament should be an important component of the force-generating step per se. Finally, our data challenge the general view that phalloidin-stabilized filaments behave as native actin filaments in their interaction with myosin. This has important implications, since phalloidin stabilization is a routine procedure in most studies of actomyosin function.
Figures


Similar articles
-
Myosin-Induced Gliding Patterns at Varied [MgATP] Unveil a Dynamic Actin Filament.Biophys J. 2016 Oct 4;111(7):1465-1477. doi: 10.1016/j.bpj.2016.08.025. Biophys J. 2016. PMID: 27705769 Free PMC article.
-
Trimethylamine N-oxide suppresses the activity of the actomyosin motor.Biochim Biophys Acta. 2012 Oct;1820(10):1597-604. doi: 10.1016/j.bbagen.2012.06.006. Epub 2012 Jun 15. Biochim Biophys Acta. 2012. PMID: 22705940
-
The excluded volume effect induced by poly(ethylene glycol) modulates the motility of actin filaments interacting with myosin.FEBS J. 2013 Nov;280(22):5875-83. doi: 10.1111/febs.12513. Epub 2013 Sep 23. FEBS J. 2013. PMID: 24004408
-
Orientation of actin monomers in moving actin filaments.Adv Exp Med Biol. 1993;332:321-8; discussion 329. doi: 10.1007/978-1-4615-2872-2_31. Adv Exp Med Biol. 1993. PMID: 8109346 Review.
-
Towards a structural understanding of the remodeling of the actin cytoskeleton.Semin Cell Dev Biol. 2020 Jun;102:51-64. doi: 10.1016/j.semcdb.2019.11.018. Epub 2019 Dec 10. Semin Cell Dev Biol. 2020. PMID: 31836290 Free PMC article. Review.
Cited by
-
Philament: A filament tracking program to quickly and accurately analyze in vitro motility assays.Biophys Rep (N Y). 2024 Jan 30;4(1):100147. doi: 10.1016/j.bpr.2024.100147. eCollection 2024 Mar 13. Biophys Rep (N Y). 2024. PMID: 38404534 Free PMC article.
-
Myosin-Induced Gliding Patterns at Varied [MgATP] Unveil a Dynamic Actin Filament.Biophys J. 2016 Oct 4;111(7):1465-1477. doi: 10.1016/j.bpj.2016.08.025. Biophys J. 2016. PMID: 27705769 Free PMC article.
-
Self-organization of motor-propelled cytoskeletal filaments at topographically defined borders.J Biomed Biotechnol. 2012;2012:647265. doi: 10.1155/2012/647265. Epub 2012 Mar 26. J Biomed Biotechnol. 2012. PMID: 22536023 Free PMC article.
-
ATLAS: Machine learning-enhanced filament analysis for the In Vitro Motility Assay.Biophys Rep (N Y). 2025 Jun 21;5(3):100221. doi: 10.1016/j.bpr.2025.100221. Online ahead of print. Biophys Rep (N Y). 2025. PMID: 40550418 Free PMC article.
-
Actomyosin based contraction: one mechanokinetic model from single molecules to muscle?J Muscle Res Cell Motil. 2016 Dec;37(6):181-194. doi: 10.1007/s10974-016-9458-0. Epub 2016 Nov 18. J Muscle Res Cell Motil. 2016. PMID: 27864648 Free PMC article.
References
-
- Huxley, A. F. 1957. Muscle structure and theories of contraction. Prog. Biophys. Biophys. Chem. 7:255–318. - PubMed
-
- Geeves, M. A., and K. C. Holmes. 2005. The molecular mechanism of muscle contraction. Adv. Protein Chem. 71:161–193. - PubMed
-
- Isambert, H., P. Venier, A. C. Maggs, A. Fattoum, R. Kassab, D. Pantaloni, and M. F. Carlier. 1995. Flexibility of actin filaments derived from thermal fluctuations. Effect of bound nucleotide, phalloidin, and muscle regulatory proteins. J. Biol. Chem. 270:11437–11444. - PubMed
-
- Orlova, A., E. Prochniewicz, and E. H. Egelman. 1995. Structural dynamics of F-actin: II. Cooperativity in structural transitions. J. Mol. Biol. 245:598–607. - PubMed
-
- Schutt, C. E., J. C. Myslik, M. D. Rozycki, N. C. Goonesekere, and U. Lindberg. 1993. The structure of crystalline profilin-β-actin. Nature. 365:810–816. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

