Cell polarity signaling in Arabidopsis
- PMID: 18837672
- PMCID: PMC2739732
- DOI: 10.1146/annurev.cellbio.23.090506.123233
Cell polarity signaling in Arabidopsis
Abstract
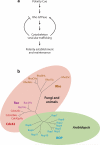
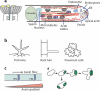
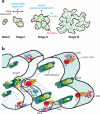
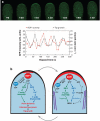
Cell polarization is intimately linked to plant development, growth, and responses to the environment. Major advances have been made in our understanding of the signaling pathways and networks that regulate cell polarity in plants owing to recent studies on several model systems, e.g., tip growth in pollen tubes, cell morphogenesis in the leaf epidermis, and polar localization of PINs. From these studies we have learned that plant cells use conserved mechanisms such as Rho family GTPases to integrate both plant-specific and conserved polarity cues and to coordinate the cytoskeketon dynamics/reorganization and vesicular trafficking required for polarity establishment and maintenance. This review focuses upon signaling mechanisms for cell polarity formation in Arabidopsis, with an emphasis on Rho GTPase signaling in polarized cell growth and how these mechanisms compare with those for cell polarity signaling in yeast and animal systems.
Figures

Similar articles
-
Arabidopsis interdigitating cell growth requires two antagonistic pathways with opposing action on cell morphogenesis.Cell. 2005 Mar 11;120(5):687-700. doi: 10.1016/j.cell.2004.12.026. Cell. 2005. PMID: 15766531
-
The ROP2 GTPase controls the formation of cortical fine F-actin and the early phase of directional cell expansion during Arabidopsis organogenesis.Plant Cell. 2002 Apr;14(4):777-94. doi: 10.1105/tpc.001537. Plant Cell. 2002. PMID: 11971134 Free PMC article.
-
A tip-localized RhoGAP controls cell polarity by globally inhibiting Rho GTPase at the cell apex.Curr Biol. 2008 Dec 23;18(24):1907-16. doi: 10.1016/j.cub.2008.11.057. Curr Biol. 2008. PMID: 19108776 Free PMC article.
-
Spatial control of plasma membrane domains: ROP GTPase-based symmetry breaking.Curr Opin Plant Biol. 2012 Dec;15(6):601-7. doi: 10.1016/j.pbi.2012.10.004. Epub 2012 Nov 20. Curr Opin Plant Biol. 2012. PMID: 23177207 Free PMC article. Review.
-
Actin polymerization processes in plant cells.Curr Opin Plant Biol. 2002 Dec;5(6):502-6. doi: 10.1016/s1369-5266(02)00300-x. Curr Opin Plant Biol. 2002. PMID: 12393012 Review.
Cited by
-
Plant cell polarity: The many facets of sidedness.Plant Physiol. 2023 Aug 31;193(1):1-5. doi: 10.1093/plphys/kiad436. Plant Physiol. 2023. PMID: 37565502 Free PMC article. No abstract available.
-
Cell surface ABP1-TMK auxin-sensing complex activates ROP GTPase signaling.Science. 2014 Feb 28;343(6174):1025-8. doi: 10.1126/science.1245125. Science. 2014. PMID: 24578577 Free PMC article.
-
The Microtubule-Associated Protein IQ67 DOMAIN5 Modulates Microtubule Dynamics and Pavement Cell Shape.Plant Physiol. 2018 Aug;177(4):1555-1568. doi: 10.1104/pp.18.00558. Epub 2018 Jul 5. Plant Physiol. 2018. PMID: 29976837 Free PMC article.
-
Phosphoinositides regulate clathrin-dependent endocytosis at the tip of pollen tubes in Arabidopsis and tobacco.Plant Cell. 2010 Dec;22(12):4031-44. doi: 10.1105/tpc.110.076760. Epub 2010 Dec 28. Plant Cell. 2010. PMID: 21189293 Free PMC article.
-
Emerging roles of small GTPases in secondary cell wall development.Front Plant Sci. 2014 Aug 26;5:428. doi: 10.3389/fpls.2014.00428. eCollection 2014. Front Plant Sci. 2014. PMID: 25206358 Free PMC article. Review.
References
-
- Aida M, Beis D, Heidstra R, Willemsen V, Blilou I, et al. The PLETHORA genes mediate patterning of the Arabidopsis root stem cell niche. Cell. 2004;119:109–20. - PubMed
-
- Basu R, Chang F. Shaping the actin cytoskeleton using microtubule tips. Curr. Opin. Cell Biol. 2007;19:88–94. - PubMed
-
- Benjamins R, Quint A, Weijers D, Hooykaas P, Offringa R. The PINOID protein kinase regulates organ development in Arabidopsis by enhancing polar auxin transport. Development. 2001;128:4057–67. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

