Rab-coupling protein coordinates recycling of alpha5beta1 integrin and EGFR1 to promote cell migration in 3D microenvironments
- PMID: 18838556
- PMCID: PMC2557049
- DOI: 10.1083/jcb.200804140
Rab-coupling protein coordinates recycling of alpha5beta1 integrin and EGFR1 to promote cell migration in 3D microenvironments
Abstract
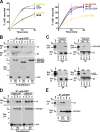
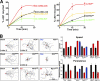
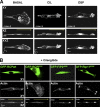
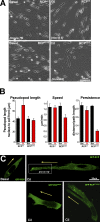
Here we show that blocking the adhesive function of alphavbeta3 integrin with soluble RGD ligands, such as osteopontin or cilengitide, promoted association of Rab-coupling protein (RCP) with alpha5beta1 integrin and drove RCP-dependent recycling of alpha5beta1 to the plasma membrane and its mobilization to dynamic ruffling protrusions at the cell front. These RCP-driven changes in alpha5beta1 trafficking led to acquisition of rapid/random movement on two-dimensional substrates and to a marked increase in fibronectin-dependent migration of tumor cells into three-dimensional matrices. Recycling of alpha5beta1 integrin did not affect its regulation or ability to form adhesive bonds with substrate fibronectin. Instead, alpha5beta1 controlled the association of EGFR1 with RCP to promote the coordinate recycling of these two receptors. This modified signaling downstream of EGFR1 to increase its autophosphorylation and activation of the proinvasive kinase PKB/Akt. We conclude that RCP provides a scaffold that promotes the physical association and coordinate trafficking of alpha5beta1 and EGFR1 and that this drives migration of tumor cells into three-dimensional matrices.
Figures




References
-
- Boettiger, D. 2007. Quantitative measurements of integrin-mediated adhesion to extracellular matrix. Methods Enzymol. 426:1–25. - PubMed
-
- Brooks, P.C., R.A. Clark, and D.A. Cheresh. 1994. Requirement of vascular integrin alpha v beta 3 for angiogenesis. Science. 264:569–571. - PubMed
-
- Caswell, P.T., and J.C. Norman. 2006. Integrin trafficking and the control of cell migration. Traffic. 7:14–21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

