TLD assessment of mouse dosimetry during microCT imaging
- PMID: 18841837
- PMCID: PMC2809703
- DOI: 10.1118/1.2959847
TLD assessment of mouse dosimetry during microCT imaging
Abstract
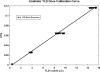
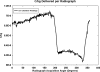
Advances in laboratory animal imaging have provided new resources for noninvasive biomedical research. Among these technologies is microcomputed tomography (microCT) which is widely used to obtain high resolution anatomic images of small animals. Because microCT utilizes ionizing radiation for image formation, radiation exposure during imaging is a concern. The objective of this study was to quantify the radiation dose delivered during a standard microCT scan. Radiation dose was measured using thermoluminescent dosimeters (TLDs), which were irradiated employing an 80 kVp x-ray source, with 0.5 mm A1 filtration and a total of 54 mA s for a full 360 deg rotation of the unit. The TLD data were validated using a 3.2 cm3 CT ion chamber probe. TLD results showed a single microCT scan air kerma of 78.0 +/- 5.0 mGy when using a poly(methylmethacrylate) (PMMA) anesthesia support module and an air kerma of 92.0 +/- 6.0 mGy without the use of the anesthesia module. The validation CT ion chamber study provided a measured radiation air kerma of 81.0 +/- 4.0 mGy and 97.0 +/- 5.0 mGy with and without the PMMA anesthesia module, respectively. Internal TLD analysis demonstrated an average mouse organ radiation absorbed dose of 76.0 +/- 5.0 mGy. The author's results have defined x-ray exposure for a routine microCT study which must be taken into consideration when performing serial molecular imaging studies involving the microCT imaging modality.
Figures



Similar articles
-
LiF:Mg,Ti TLD response as a function of photon energy for moderately filtered x-ray spectra in the range of 20-250 kVp relative to 60Co.Med Phys. 2008 May;35(5):1859-69. doi: 10.1118/1.2898137. Med Phys. 2008. PMID: 18561661
-
Response of LiF:Mg,Ti thermoluminescent dosimeters at photon energies relevant to the dosimetry of brachytherapy (<1 MeV).Med Phys. 2011 Oct;38(10):5539-50. doi: 10.1118/1.3633892. Med Phys. 2011. PMID: 21992372
-
Absorbed radiation dose of the female breast during diagnostic multidetector chest CT and dose reduction with a tungsten-antimony composite breast shield: preliminary results.Clin Radiol. 2008 Mar;63(3):278-88. doi: 10.1016/j.crad.2007.07.029. Epub 2007 Dec 4. Clin Radiol. 2008. PMID: 18275868
-
Studying the dose level for different X-ray energy conventional radiography by TLD-100.Appl Radiat Isot. 2022 Mar;181:110066. doi: 10.1016/j.apradiso.2021.110066. Epub 2021 Dec 18. Appl Radiat Isot. 2022. PMID: 34968881 Review.
-
High resolution X-ray computed tomography: an emerging tool for small animal cancer research.Neoplasia. 2000 Jan-Apr;2(1-2):62-70. doi: 10.1038/sj.neo.7900069. Neoplasia. 2000. PMID: 10933069 Free PMC article. Review.
Cited by
-
X-ray phase-contrast tomography with a compact laser-driven synchrotron source.Proc Natl Acad Sci U S A. 2015 May 5;112(18):5567-72. doi: 10.1073/pnas.1500938112. Epub 2015 Apr 20. Proc Natl Acad Sci U S A. 2015. PMID: 25902493 Free PMC article.
-
Targeted multi-pinhole SPECT.Eur J Nucl Med Mol Imaging. 2011 Mar;38(3):552-61. doi: 10.1007/s00259-010-1637-4. Epub 2010 Nov 10. Eur J Nucl Med Mol Imaging. 2011. PMID: 21063706 Free PMC article.
-
Dosimetry in Micro-computed Tomography: a Review of the Measurement Methods, Impacts, and Characterization of the Quantum GX Imaging System.Mol Imaging Biol. 2017 Aug;19(4):499-511. doi: 10.1007/s11307-016-1026-x. Mol Imaging Biol. 2017. PMID: 27957647 Free PMC article. Review.
-
X-ray phase-contrast CT of a pancreatic ductal adenocarcinoma mouse model.PLoS One. 2013;8(3):e58439. doi: 10.1371/journal.pone.0058439. Epub 2013 Mar 11. PLoS One. 2013. PMID: 23536795 Free PMC article.
-
Low-Dose Imaging in a New Preclinical Total-Body PET/CT Scanner.Front Med (Lausanne). 2019 May 3;6:88. doi: 10.3389/fmed.2019.00088. eCollection 2019. Front Med (Lausanne). 2019. PMID: 31131277 Free PMC article.
References
-
- Arguello F., Baggs R. B., and Frantz C. N., “A murine model of experimental metastasis to bone and bone marrow,” Cancer Res. CNREA8 48, 6876–6881 (1988). - PubMed
-
- Yang M., Jiang P., An Z., Baranov E., Li L., Hasegawa S., Al-Tuwaijri M., Chishima T., Shimada H., Moossa A. R., and Hoffman R. M., “Genetically fluorescent melanoma bone and organ metastasis models,” Clin. Cancer Res. ZZZZZZ 5, 3549–3559 (1999). - PubMed
-
- Ray P., Wu A. M., and Gambhir S. S., “Optical bioluminescence and positron emission tomography imaging of a novel fusion reporter gene in tumor xenografts of living mice,” Cancer Res. CNREA8 63, 1160–1165 (2003). - PubMed
-
- Shrayer D. P., Bogaars H., Wolf S. F., Hearing V. J., and Wanebo H. J., “A new mouse model of experimental melanoma for vaccine and lymphokine therapy,” Int. J. Oncol. ZZZZZZ 13, 361–364 (1998). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

