Different structural changes occur in blue- and green-proteorhodopsins during the primary photoreaction
- PMID: 18842006
- PMCID: PMC3631532
- DOI: 10.1021/bi800945t
Different structural changes occur in blue- and green-proteorhodopsins during the primary photoreaction
Abstract
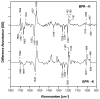
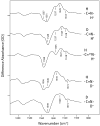
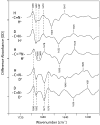
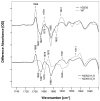
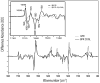
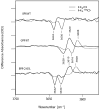
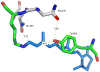
We examine the structural changes during the primary photoreaction in blue-absorbing proteorhodopsin (BPR), a light-driven retinylidene proton pump, using low-temperature FTIR difference spectroscopy. Comparison of the light-induced BPR difference spectrum recorded at 80 K to that of green-absorbing proteorhodopsin (GPR) reveals that there are several differences in the BPR and GPR primary photoreactions despite the similar structure of the retinal chromophore and all-trans --> 13-cis isomerization. Strong bands near 1700 cm(-1) assigned previously to a change in hydrogen bonding of Asn230 in GPR are still present in BPR. However, additional bands in the same region are assigned on the basis of site-directed mutagenesis to changes occurring in Gln105. In the amide II region, bands are assigned on the basis of total (15)N labeling to structural changes of the protein backbone, although no such bands were previously observed for GPR. A band at 3642 cm(-1) in BPR, assigned to the OH stretching mode of a water molecule on the basis of H2(18)O substitution, appears at a different frequency than a band at 3626 cm(-1) previously assigned to a water molecule in GPR. However, the substitution of Gln105 for Leu105 in BPR leads to the appearance of both bands at 3642 and 3626 cm(-1), indicating the waters assigned in BPR and GPR exist in separate distinct locations and can coexist in the GPR-like Q105L mutant of BPR. These results indicate that there exist significant differences in the conformational changes occurring in these two types proteorhodopsin during the initial photoreaction despite their similar chromophore structures, which might reflect a different arrangement of water in the active site as well as substitution of a hydrophilic for hydrophobic residue at residue 105.
Figures
References
-
- Beja O, Aravind L, Koonin EV, Suzuki MT, Hadd A, Nguyen LP, Jovanovich SB, Gates CM, Feldman RA, Spudich JL, Spudich EN, DeLong EF. Bacterial rhodopsin: evidence for a new type of phototrophy in the sea. Science. 2000;289:1902–1906. - PubMed
-
- Rusch DB, Halpern AL, Sutton G, Heidelberg KB, Williamson S, Yooseph S, Wu D, Eisen JA, Hoffman JM, Remington K, Beeson K, Tran B, Smith H, Baden-Tillson H, Stewart C, Thorpe J, Freeman J, Andrews-Pfannkoch C, Venter JE, Li K, Kravitz S, Heidelberg JF, Utterback T, Rogers Y-H, Falcn LI, Souza V, Bonilla-Rosso G, Eguiarte LE, Karl DM, Sathyendranath S, Platt T, Bermingham E, Gallardo V, Tamayo-Castillo G, Ferrari MR, Strausberg RL, Nealson K, Friedman R, Frazier M, Venter JC. The Sorcerer II Global Ocean Sampling Expedition: Northwest Atlantic through Eastern Tropical Pacific. PLoS Biology. 2007;5:e77. - PMC - PubMed
-
- Spudich JL, Jung K-H. Microbial Rhodopsins: Phylogenetic and Functional Diversity. 2005. pp. 1–24.
-
- Venter JC, Remington K, Heidelberg JF, Halpern AL, Rusch D, Eisen JA, Wu D, Paulsen I, Nelson KE, Nelson W, Fouts DE, Levy S, Knap AH, Lomas MW, Nealson K, White O, Peterson J, Hoffman J, Parsons R, Baden-Tillson H, Pfannkoch C, Rogers YH, Smith HO. Environmental genome shotgun sequencing of the Sargasso Sea. Science. 2004;304:66–74. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

