Proton-coupled electron transfer in solution, proteins, and electrochemistry
- PMID: 18842015
- PMCID: PMC2720037
- DOI: 10.1021/jp805876e
Proton-coupled electron transfer in solution, proteins, and electrochemistry
Abstract
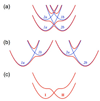
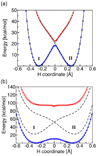
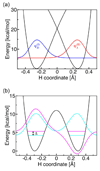
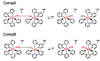
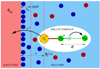
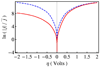
Recent advances in the theoretical treatment of proton-coupled electron transfer (PCET) reactions are reviewed. These reactions play an important role in a wide range of biological processes, as well as in fuel cells, solar cells, chemical sensors, and electrochemical devices. A unified theoretical framework has been developed to describe both sequential and concerted PCET, as well as hydrogen atom transfer (HAT). A quantitative diagnostic has been proposed to differentiate between HAT and PCET in terms of the degree of electronic nonadiabaticity, where HAT corresponds to electronically adiabatic proton transfer and PCET corresponds to electronically nonadiabatic proton transfer. In both cases, the overall reaction is typically vibronically nonadiabatic. A series of rate constant expressions have been derived in various limits by describing the PCET reactions in terms of nonadiabatic transitions between electron-proton vibronic states. These expressions account for the solvent response to both electron and proton transfer and the effects of the proton donor-acceptor vibrational motion. The solvent and protein environment can be represented by a dielectric continuum or described with explicit molecular dynamics. These theoretical treatments have been applied to numerous PCET reactions in solution and proteins. Expressions for heterogeneous rate constants and current densities for electrochemical PCET have also been derived and applied to model systems.
Figures




References
-
- Roberts JA, Kirby JP, Nocera DG. Journal of the American Chemical Society. 1995;117:8051.
-
- Reece SY, Nocera DG. Journal of the American Chemical Society. 2005;127:9448. - PubMed
-
- Rosenthal J, Nocera DG. Accounts of Chemical Research. 2007;40:543. - PubMed
-
- Irebo T, Reece SY, Sjodin M, Nocera DG, Hammarstrom L. Journal of the American Chemical Society. 2007;129:15462. - PubMed
-
- Sjodin M, Styring S, Akermark B, Sun L, Hammarstrom L. Journal of the American Chemical Society. 2000;122:3932.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

