Regulation of beta-amyloid levels in the brain of cholesterol-fed rabbit, a model system for sporadic Alzheimer's disease
- PMID: 18845178
- PMCID: PMC2629555
- DOI: 10.1016/j.mad.2008.09.002
Regulation of beta-amyloid levels in the brain of cholesterol-fed rabbit, a model system for sporadic Alzheimer's disease
Abstract
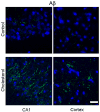
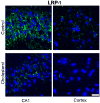
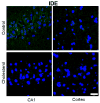
Accumulation of beta-amyloid (Abeta) peptide in the brain is a major hallmark of Alzheimer's disease (AD). Hypercholesterolemia is a risk factor for AD and has been shown by laboratory studies to cause Abeta accumulation. Abeta levels in the brain are governed by its generation from amyloid precursor protein by beta-secretase (BACE1), degradation by the insulin degrading enzyme (IDE), clearance from the brain by the low density lipoprotein receptor-related protein (LRP-1), and transport from circulation into the brain by receptor for advanced glycation end products (RAGE). However, the mechanisms by which hypercholesterolemia causes Abeta accumulation in the brain and contributes to the pathogenesis of AD are still to be determined. In the present study, we determined the extent to which hypercholesterolemia-induced Abeta accumulation is associated with alterations in BACE1, IDE, LRP-1, and RAGE expression levels. We show that hypercholesterolemia increases Abeta production, an effect that is associated with increased levels of BACE1 and RAGE and reduced levels of IDE and LRP-1. These results suggest that reducing Abeta accumulation in the brain may require strategies that combine reduction of generation and transport of Abeta in addition to acceleration of degradation and clearance of this peptide.
Figures






Similar articles
-
Thymoquinone-rich fraction nanoemulsion (TQRFNE) decreases Aβ40 and Aβ42 levels by modulating APP processing, up-regulating IDE and LRP1, and down-regulating BACE1 and RAGE in response to high fat/cholesterol diet-induced rats.Biomed Pharmacother. 2017 Nov;95:780-788. doi: 10.1016/j.biopha.2017.08.074. Epub 2017 Sep 8. Biomed Pharmacother. 2017. PMID: 28892789
-
Effect of High Cholesterol Regulation of LRP1 and RAGE on Aβ Transport Across the Blood-Brain Barrier in Alzheimer's Disease.Curr Alzheimer Res. 2021;18(5):428-442. doi: 10.2174/1567205018666210906092940. Curr Alzheimer Res. 2021. PMID: 34488598
-
Impact of age, hypercholesterolemia, and the vitamin D receptor on brain endogenous β-amyloid peptide accumulation in mice.Biopharm Drug Dispos. 2021 Sep;42(8):372-388. doi: 10.1002/bdd.2297. Epub 2021 Aug 18. Biopharm Drug Dispos. 2021. PMID: 34219248
-
Current Updates on the Regulation of Beta-Secretase Movement as a Potential Restorative Focus for Management of Alzheimer's Disease.Protein Pept Lett. 2019;26(8):579-587. doi: 10.2174/0929866526666190405125334. Protein Pept Lett. 2019. PMID: 30950339 Review.
-
Clearance of amyloid-beta peptide across the blood-brain barrier: implication for therapies in Alzheimer's disease.CNS Neurol Disord Drug Targets. 2009 Mar;8(1):16-30. doi: 10.2174/187152709787601867. CNS Neurol Disord Drug Targets. 2009. PMID: 19275634 Free PMC article. Review.
Cited by
-
Amyloid Beta Peptides Affect Pregnenolone and Pregnenolone Sulfate Levels in PC-12 and SH-SY5Y Cells Depending on Cholesterol.Neurochem Res. 2016 Jul;41(7):1700-12. doi: 10.1007/s11064-016-1886-0. Epub 2016 Mar 26. Neurochem Res. 2016. PMID: 27017511
-
The effects of cholesterol on learning and memory.Neurosci Biobehav Rev. 2010 Jul;34(8):1366-79. doi: 10.1016/j.neubiorev.2010.04.010. Epub 2010 May 12. Neurosci Biobehav Rev. 2010. PMID: 20470821 Free PMC article. Review.
-
Cholesterol as a key player in amyloid β-mediated toxicity in Alzheimer's disease.Front Mol Neurosci. 2022 Aug 25;15:937056. doi: 10.3389/fnmol.2022.937056. eCollection 2022. Front Mol Neurosci. 2022. PMID: 36090253 Free PMC article. Review.
-
Association studies of several cholesterol-related genes (ABCA1, CETP and LIPC) with serum lipids and risk of Alzheimer's disease.Lipids Health Dis. 2012 Nov 26;11:163. doi: 10.1186/1476-511X-11-163. Lipids Health Dis. 2012. PMID: 23181436 Free PMC article.
-
Role of cholesterol in APP metabolism and its significance in Alzheimer's disease pathogenesis.Mol Neurobiol. 2013 Feb;47(1):37-63. doi: 10.1007/s12035-012-8337-y. Epub 2012 Sep 16. Mol Neurobiol. 2013. PMID: 22983915 Review.
References
-
- Affholter JA, Hsieh CL, Francke U, Roth RA. Insulin degrading enzyme: stable expression of the human complementary DNA, characterization of its protein product, and chromosomal mapping of the human and mouse genes. Molecular Endocrinology. 1990;4(8):1125–1135. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

