Transforming growth factor beta 1 and hyaluronan oligomers synergistically enhance elastin matrix regeneration by vascular smooth muscle cells
- PMID: 18847364
- PMCID: PMC2810272
- DOI: 10.1089/ten.tea.2008.0040
Transforming growth factor beta 1 and hyaluronan oligomers synergistically enhance elastin matrix regeneration by vascular smooth muscle cells
Abstract
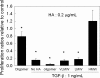
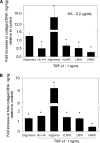
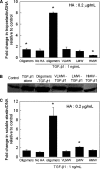
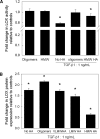
Elastin is a vital structural and regulatory matrix protein that plays an important role in conferring elasticity to blood vessel wall. Previous tissue engineering approaches to regenerate elastin in situ or within tissue engineering constructs are curtailed by innate poor elastin synthesis potential by adult vascular smooth muscle cells (SMCs). Currently, we seek to develop cellular cues to enhance tropoelastin synthesis and improve elastin matrix yield, stability, and ultrastructure. Our earlier studies attest to the elastogenic utility of hyaluronan (HA)-based cellular cues, though their effects are fragment size dependent and dose dependent, with HA oligomers deemed most elastogenic. We presently show transforming growth factor beta 1 (TGF-beta1) and HA oligomers, when provided concurrently, to synergistically and dramatically improve elastin matrix regeneration by adult vascular SMCs. Together, these cues suppress SMC proliferation, enhance synthesis of tropoelastin (8-fold) and matrix elastin protein (5.5-fold), and also improve matrix elastin yield (45% of total elastin vs. 10% for nonadditive controls), possibly by more efficient recruitment of tropoelastin for crosslinking. The density of desmosine crosslinks within the elastin matrix was itself attenuated, although the cues together modestly increased production and activity of the elastin crosslinking enzyme, lysyl oxidase. TGF-beta1 and HA oligomers together induced much greater assembly of mature elastin fibers than they did separately, and did not induce matrix calcification. The present outcomes might be great utility to therapeutic regeneration of elastin matrix networks in situ within elastin-compromised vessels, and within tissue-engineered vascular graft replacements.
Figures


Similar articles
-
Benefits of concurrent delivery of hyaluronan and IGF-1 cues to regeneration of crosslinked elastin matrices by adult rat vascular cells.J Tissue Eng Regen Med. 2008 Mar-Apr;2(2-3):106-16. doi: 10.1002/term.70. J Tissue Eng Regen Med. 2008. PMID: 18338830
-
Utility of hyaluronan oligomers and transforming growth factor-beta1 factors for elastic matrix regeneration by aneurysmal rat aortic smooth muscle cells.Tissue Eng Part A. 2009 Nov;15(11):3247-60. doi: 10.1089/ten.tea.2008.0593. Tissue Eng Part A. 2009. PMID: 19374489 Free PMC article.
-
Elastogenic effects of exogenous hyaluronan oligosaccharides on vascular smooth muscle cells.Biomaterials. 2006 Nov;27(33):5698-707. doi: 10.1016/j.biomaterials.2006.07.020. Epub 2006 Aug 8. Biomaterials. 2006. PMID: 16899292
-
Elastin: relation of protein and gene structure to disease.Lab Invest. 1984 Dec;51(6):605-23. Lab Invest. 1984. PMID: 6150137 Review.
-
Tropoelastin.Int J Biochem Cell Biol. 2009 Mar;41(3):494-7. doi: 10.1016/j.biocel.2008.03.017. Epub 2008 Apr 1. Int J Biochem Cell Biol. 2009. PMID: 18468477 Review.
Cited by
-
Induced Regenerative Elastic Matrix Repair in LOXL1 Knockout Mouse Cell Cultures: Towards Potential therapy for Pelvic Organ Prolapse.J Tissue Sci Eng. 2012;3(3):120. doi: 10.4172/2157-7552.1000120. Epub 2012 Sep 28. J Tissue Sci Eng. 2012. PMID: 30854248 Free PMC article.
-
JNK2 Gene Silencing for Elastic Matrix Regenerative Repair.Tissue Eng Part A. 2022 Mar;28(5-6):239-253. doi: 10.1089/ten.TEA.2020.0221. Tissue Eng Part A. 2022. PMID: 34409851 Free PMC article.
-
Impact of pre-existing elastic matrix on TGFβ1 and HA oligomer-induced regenerative elastin repair by rat aortic smooth muscle cells.J Tissue Eng Regen Med. 2011 Feb;5(2):85-96. doi: 10.1002/term.286. J Tissue Eng Regen Med. 2011. PMID: 20653044 Free PMC article.
-
Composition of intraperitoneally implanted electrospun conduits modulates cellular elastic matrix generation.Acta Biomater. 2014 Jan;10(1):163-72. doi: 10.1016/j.actbio.2013.08.042. Epub 2013 Sep 7. Acta Biomater. 2014. PMID: 24016842 Free PMC article.
-
Hyaluronan - a functional and structural sweet spot in the tissue microenvironment.Front Immunol. 2015 May 15;6:231. doi: 10.3389/fimmu.2015.00231. eCollection 2015. Front Immunol. 2015. PMID: 26029216 Free PMC article. Review.
References
-
- Davidson J.M. Giro M.G. Regulation of matrix accumulation. In: Mecham R.P., editor. Biology of the extracellular matrix. NY: Academic Press; 1986. pp. 178–216.
-
- Kielty C.M. Sherratt M.J. Shuttleworth C.A. Elastic fibres. J Cell Sci. 2002;115:2817. - PubMed
-
- Robert L. Jacob M.P. Fulop T. Cell–elastin interaction and signaling. Pathol Biol. 2005;53:399. - PubMed
-
- Rodgers U.R. Weiss A.S. Cellular interactions with elastin. Pathol Biol. 2005;53:390. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

