Stabilized beta-catenin functions through TCF/LEF proteins and the Notch/RBP-Jkappa complex to promote proliferation and suppress differentiation of neural precursor cells
- PMID: 18852283
- PMCID: PMC2593432
- DOI: 10.1128/MCB.01962-07
Stabilized beta-catenin functions through TCF/LEF proteins and the Notch/RBP-Jkappa complex to promote proliferation and suppress differentiation of neural precursor cells
Abstract
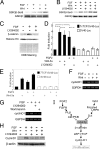
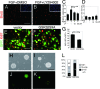
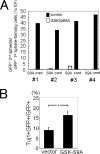
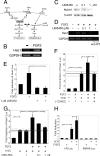
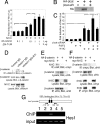
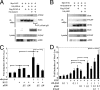
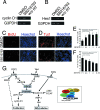
The proliferation and differentiation of neural precursor cells are mutually exclusive during brain development. Despite its importance for precursor cell self renewal, the molecular linkage between these two events has remained unclear. Fibroblast growth factor 2 (FGF2) promotes neural precursor cell proliferation and concurrently inhibits their differentiation, suggesting a cross talk between proliferation and differentiation signaling pathways downstream of the FGF receptor. We demonstrate that FGF2 signaling through phosphatidylinositol 3 kinase activation inactivates glycogen synthase kinase 3beta (GSK3beta) and leads to the accumulation of beta-catenin in a manner different from that in the Wnt canonical pathway. The nuclear accumulated beta-catenin leads to cell proliferation by activating LEF/TCF transcription factors and concurrently inhibits neuronal differentiation by potentiating the Notch1-RBP-Jkappa signaling pathway. beta-Catenin and the Notch1 intracellular domain form a molecular complex with the promoter region of the antineurogenic hes1 gene, allowing its expression. This signaling interplay is especially essential for neural stem cell maintenance, since the misexpression of dominant-active GSK3beta completely inhibits the self renewal of neurosphere-forming stem cells and prompts their neuronal differentiation. Thus, the GSK3beta/beta-catenin signaling axis regulated by FGF and Wnt signals plays a pivotal role in the maintenance of neural stem/precursor cells by linking the cell proliferation to the inhibition of differentiation.
Figures
References
-
- Alves-Guerra, M. C., C. Ronchini, and A. J. Capobianco. 2007. Mastermind-like 1 is a specific coactivator of beta-catenin transcription activation and is essential for colon carcinoma cell survival. Cancer Res. 678690-8698. - PubMed
-
- Amit, M., M. K. Carpenter, M. S. Inokuma, C. P. Chiu, C. P. Harris, M. A. Waknitz, J. Itskovitz-Eldor, and J. A. Thomson. 2000. Clonally derived human embryonic stem cell lines maintain pluripotency and proliferative potential for prolonged periods of culture. Dev. Biol. 227271-278. - PubMed
-
- Andjelkoviæ, M., D. R. Alessi, R. Meier, A. Fernandez, N. J. Lamb, M. Frech, P. Cron, P. Cohen, J. M. Lucocq, and B. A. Hemmings. 1997. Role of translocation in the activation and function of protein kinase B. J. Biol. Chem. 27231515-31524. - PubMed
-
- Belham, C., S. Wu, and J. Avruch. 1999. Intracellular signalling: PDK1-a kinase at the hub of things. Curr. Biol. 9R93-R96. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous
