M-CSF signals through the MAPK/ERK pathway via Sp1 to induce VEGF production and induces angiogenesis in vivo
- PMID: 18852899
- PMCID: PMC2566603
- DOI: 10.1371/journal.pone.0003405
M-CSF signals through the MAPK/ERK pathway via Sp1 to induce VEGF production and induces angiogenesis in vivo
Abstract
Background: M-CSF recruits mononuclear phagocytes which regulate processes such as angiogenesis and metastases in tumors. VEGF is a potent activator of angiogenesis as it promotes endothelial cell proliferation and new blood vessel formation. Previously, we reported that in vitro M-CSF induces the expression of biologically-active VEGF from human monocytes.
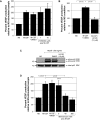
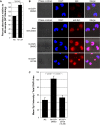
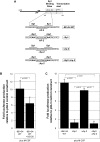
Methodology and results: In this study, we demonstrate the molecular mechanism of M-CSF-induced VEGF production. Using a construct containing the VEGF promoter linked to a luciferase reporter, we found that a mutation reducing HIF binding to the VEGF promoter had no significant effect on luciferase production induced by M-CSF stimulation. Further analysis revealed that M-CSF induced VEGF through the MAPK/ERK signaling pathway via the transcription factor, Sp1. Thus, inhibition of either ERK or Sp1 suppressed M-CSF-induced VEGF at the mRNA and protein level. M-CSF also induced the nuclear localization of Sp1, which was blocked by ERK inhibition. Finally, mutating the Sp1 binding sites within the VEGF promoter or inhibiting ERK decreased VEGF promoter activity in M-CSF-treated human monocytes. To evaluate the biological significance of M-CSF induced VEGF production, we used an in vivo angiogenesis model to illustrate the ability of M-CSF to recruit mononuclear phagocytes, increase VEGF levels, and enhance angiogenesis. Importantly, the addition of a neutralizing VEGF antibody abolished M-CSF-induced blood vessel formation.
Conclusion: These data delineate an ERK- and Sp1-dependent mechanism of M-CSF induced VEGF production and demonstrate for the first time the ability of M-CSF to induce angiogenesis via VEGF in vivo.
Conflict of interest statement
Figures

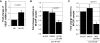

Similar articles
-
Dimethylarginine dimethylaminohydrolase 2 increases vascular endothelial growth factor expression through Sp1 transcription factor in endothelial cells.Arterioscler Thromb Vasc Biol. 2006 Jul;26(7):1488-94. doi: 10.1161/01.ATV.0000219615.88323.b4. Epub 2006 Mar 30. Arterioscler Thromb Vasc Biol. 2006. PMID: 16574895
-
Cysteinyl-leukotrienes induce vascular endothelial growth factor production in human monocytes and bronchial smooth muscle cells.Clin Exp Allergy. 2011 Feb;41(2):204-17. doi: 10.1111/j.1365-2222.2010.03653.x. Epub 2010 Dec 1. Clin Exp Allergy. 2011. PMID: 21121979
-
Low-power laser irradiation (LPLI) promotes VEGF expression and vascular endothelial cell proliferation through the activation of ERK/Sp1 pathway.Cell Signal. 2012 Jun;24(6):1116-25. doi: 10.1016/j.cellsig.2012.01.013. Epub 2012 Feb 2. Cell Signal. 2012. PMID: 22326662
-
Macrophage colony stimulating factor: not just for macrophages anymore! A gateway into complex biologies.Int Immunopharmacol. 2008 Oct;8(10):1354-76. doi: 10.1016/j.intimp.2008.04.016. Epub 2008 Jun 2. Int Immunopharmacol. 2008. PMID: 18687298 Review.
-
Redox regulation of the VEGF signaling path and tissue vascularization: Hydrogen peroxide, the common link between physical exercise and cutaneous wound healing.Free Radic Biol Med. 2008 Jan 15;44(2):180-92. doi: 10.1016/j.freeradbiomed.2007.01.025. Epub 2007 Jan 19. Free Radic Biol Med. 2008. PMID: 18191754 Review.
Cited by
-
Inhibition of Specificity Protein 1 Is Involved in Phloretin-Induced Suppression of Prostate Cancer.Biomed Res Int. 2020 Aug 10;2020:1358674. doi: 10.1155/2020/1358674. eCollection 2020. Biomed Res Int. 2020. PMID: 32851058 Free PMC article.
-
Antiangiogenic Potential of Troxerutin and Chitosan Loaded Troxerutin on Chorioallantoic Membrane Model.Biomed Res Int. 2023 May 23;2023:5956154. doi: 10.1155/2023/5956154. eCollection 2023. Biomed Res Int. 2023. PMID: 37260851 Free PMC article.
-
Batf3 and Id2 have a synergistic effect on Irf8-directed classical CD8α+ dendritic cell development.J Immunol. 2013 Dec 15;191(12):5993-6001. doi: 10.4049/jimmunol.1203541. Epub 2013 Nov 13. J Immunol. 2013. PMID: 24227775 Free PMC article.
-
Adiponectin upregulates prolyl-4-hydroxylase α1 expression in interleukin 6-stimulated human aortic smooth muscle cells by regulating ERK 1/2 and Sp1.PLoS One. 2011;6(7):e22819. doi: 10.1371/journal.pone.0022819. Epub 2011 Jul 29. PLoS One. 2011. PMID: 21829524 Free PMC article.
-
Kiperin Postbiotic Supplement-Enhanced Bacterial Supernatants Promote Fibroblast Function: Implications for Regenerative Medicine.Biomedicines. 2025 Jun 10;13(6):1430. doi: 10.3390/biomedicines13061430. Biomedicines. 2025. PMID: 40564148 Free PMC article.
References
-
- Kelley TW, Graham MM, Doseff AI, Pomerantz RW, Lau SM, et al. Macrophage colony-stimulating factor promotes cell survival through Akt/protein kinase B. J Biol Chem. 1999;274:26393–26398. doi:10.1074/jbc.274.37.26393. PMid:10473597. - PubMed
-
- Hamilton JA, Vairo G, Lingelbach SR. Activation and proliferation signals in murine macrophages: Stimulation of glucose uptake by hemopoietic growth factors and other agents. J Cell Physiol. 1988;134:405–412. doi:10.1002/jcp.1041340311. PMid:2832422. - PubMed
-
- Junttila I, Bourette RP, Rohrschneider LR, Silvennoinen O. M-CSF induced differentiation of myeloid precursor cells involves activation of PKC-δ and expression of pkare. J Leukoc Biol. 2003;73:281–288. doi:10.1189/jlb.0702359 PMid:12554805. - PubMed
-
- Falkenburg JH, Harrington MA, Walsh WK, Daub R, Broxmeyer HE. Gene-expression and release of macrophage-colony stimulating factor in quiescent and proliferating fibroblasts. effects of serum, fibroblast growth-promoting factors, and IL-1. J Immunol. 1990;144:4657–4662. - PubMed
-
- Misago M, Tsukada J, Ogawa R, Kikuchi M, Hanamura T, et al. Enhancing effects of IL-2 on M-CSF production by human peripheral blood monocytes. Int J Hematol. 1993;58:43–51. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

