Identification of a new JNK inhibitor targeting the JNK-JIP interaction site
- PMID: 18922779
- PMCID: PMC2567907
- DOI: 10.1073/pnas.0805677105
Identification of a new JNK inhibitor targeting the JNK-JIP interaction site
Abstract
JNK is a stress-activated protein kinase that modulates pathways implicated in a variety of disease states. JNK-interacting protein-1 (JIP1) is a scaffolding protein that enhances JNK signaling by creating a proximity effect between JNK and upstream kinases. A minimal peptide region derived from JIP1 is able to inhibit JNK activity both in vitro and in cell. We report here a series of small molecules JIP1 mimics that function as substrate competitive inhibitors of JNK. One such compound, BI-78D3, dose-dependently inhibits the phosphorylation of JNK substrates both in vitro and in cell. In animal studies, BI-78D3 not only blocks JNK dependent Con A-induced liver damage but also restores insulin sensitivity in mouse models of type 2 diabetes. Our findings open the way for the development of protein kinase inhibitors targeting substrate specific docking sites, rather than the highly conserved ATP binding sites. In view of its favorable inhibition profile, selectivity, and ability to function in the cellular milieu and in vivo, BI-78D3 represents not only a JNK inhibitor, but also a promising stepping stone toward the development of an innovative class of therapeutics.
Conflict of interest statement
Conflict of interest statement: The series of compounds discovered were licensed to Syndexa Pharmaceuticals. M.P. and M.K. are shareholders and consultants for Syndexa Pharmaceuticals.
Figures
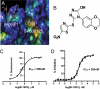
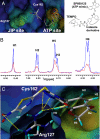

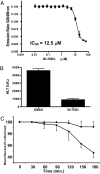
Comment in
-
Covalent JNK inhibitors?Proc Natl Acad Sci U S A. 2009 Feb 24;106(8):E18; author reply E19. doi: 10.1073/pnas.0812246106. Epub 2009 Feb 9. Proc Natl Acad Sci U S A. 2009. PMID: 19204298 Free PMC article. No abstract available.
References
-
- Derijard B, et al. JNK1: A protein kinase stimulated by UV light and Ha-Ras that binds and phosphorylates the c-Jun activation domain. Cell. 1994;76:1025–1037. - PubMed
-
- Kallunki T, et al. JNK2 contains a specificity-determining region responsible for efficient c-Jun binding and phosphorylation. Genes Dev. 1994;8:2996–3007. - PubMed
-
- Kyriakis JM, Avruch J. pp54 microtubule-associated protein 2 kinase. A novel serine/threonine protein kinase regulated by phosphorylation and stimulated by poly-L-lysine. J Biol Chem. 1990;265:17355–17363. - PubMed
-
- Martin JH, Mohit AA, Miller CA. Developmental expression in the mouse nervous system of the p493F12 SAP kinase. Brain Res Mol Brain Res. 1996;35:47–57. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Research Materials

