A novel dietary flavonoid fisetin inhibits androgen receptor signaling and tumor growth in athymic nude mice
- PMID: 18922931
- PMCID: PMC2954499
- DOI: 10.1158/0008-5472.CAN-08-0240
A novel dietary flavonoid fisetin inhibits androgen receptor signaling and tumor growth in athymic nude mice
Abstract
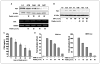
Androgen receptor (AR)-mediated signaling plays an important role in the development and progression of prostate cancer (PCa). Hormonal therapies, mainly with combinations of antiandrogens and androgen deprivation, are the mainstay treatment for advanced disease. However, emergence of androgen resistance largely due to inefficient antihormone action limits their therapeutic usefulness. Here, we report that fisetin, a novel dietary flavonoid, acts as a novel AR ligand by competing with the high-affinity androgen to interact with the ligand binding domain of AR. We show that this physical interaction results in substantial decrease in AR stability and decrease in amino-terminal/carboxyl-terminal (N-C) interaction of AR. This results in blunting of AR-mediated transactivation of target genes including prostate-specific antigen (PSA). In addition, treatment of LNCaP cells with fisetin decreased AR protein levels, in part, by decreasing its promoter activity and by accelerating its degradation. Fisetin also synergized with Casodex in inducing apoptosis in LNCaP cells. Treatment with fisetin in athymic nude mice implanted with AR-positive CWR22Rupsilon1 human PCa cells resulted in inhibition of tumor growth and reduction in serum PSA levels. These data identify fisetin as an inhibitor of AR signaling axis and suggest that it could be a useful chemopreventive and chemotherapeutic agent to delay progression of PCa.
Conflict of interest statement
No potential conflicts of interest were disclosed.
Figures




References
-
- Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics, 2007. CA Cancer J Clin. 2007;57:43–66. - PubMed
-
- Reddy GP, Barrack ER, Dou QP, et al. Regulatory processes affecting androgen receptor expression, stability, and function: potential targets to treat hormone-refractory prostate cancer. J Cell Biochem. 2006;98:1408–23. - PubMed
-
- Hsieh TC, Wu JM. Ethanolic extracts of herbal supplement Equiguardtrade mark suppress growth and control gene expression in CWR22Rv1 cells representing the transition of prostate cancer from androgen dependence to hormone refractory status. Int J Oncol. 2008;32:209–19. - PubMed
-
- Syed DN, Khan N, Afaq F, Mukhtar H. Chemoprevention of prostate cancer through dietary agents: progress and promise. Cancer Epidemiol Biomarkers Prev. 2007;16:2193–203. - PubMed
-
- Adhami VM, Mukhtar H. Anti-oxidants from green tea and pomegranate for chemoprevention of prostate cancer. Mol Biotechnol. 2007;37:52–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

