Abnormal glyceraldehyde-3-phosphate dehydrogenase binding and glycolytic flux in Autosomal Dominant Polycystic Kidney Disease after a mild oxidative stress
- PMID: 18923744
- PMCID: PMC2504404
Abnormal glyceraldehyde-3-phosphate dehydrogenase binding and glycolytic flux in Autosomal Dominant Polycystic Kidney Disease after a mild oxidative stress
Abstract
Aim: The aim of this study was, a) to investigate the effect of mild oxidative stress on glycolytic flux and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) binding in erythrocytes from patients with autosomal dominant polycystic kidney disease (ADPKD), and b) to examine whether the modulation of GAPDH-binding to the red cell membrane leads to changes in glycolytic flux.
Patients and methods: The rate of lactate production in intact erythrocytes and the GAPDH/actin ratio in erythrocyte ghost membranes were measured before and after treating cells with t-butyl hydroperoxide or N-ethylmaleimide (NEM) in 13 ADPKD patients and 12 controls.
Results: t-bytyl hydro-peroxide had a significant effect on both lactate production and GAPDH/actin ratio in healthy subjects, but it had essentially no effect on ADPKD patients in which both parameters already resembled those of the peroxide-treated controls. NEM treatment after 300 sec had a very significant effect on both lactate production and GAPDH/actin ratio in both patient and control cells. However, after 10 sec the effect on GAPDH/actin ratio was only significant in the erythrocytes of ADPKD patients. In every experiment glycolytic lactate production correlated negatively with membrane-bound GAPDH/actin ratio.
Conclusions: We conclude that glycolytic flux and GAPDH binding in erythrocytes from ADPKD patients respond abnormally to both a mild oxidative stress and brief exposure to NEM.
Keywords: autosomal dominant polycystic kidney disease; glyceraldehyde-3-phosphate dehydrogenase; glycolysis; oxidative stress.
Figures
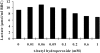
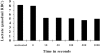
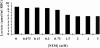
References
-
- Gabow PA. Autosomal dominant polycystic kidney disease. N Engl J Med. 1993;329:332–342. - PubMed
-
- Maser RL, Magenheimer BS, Calvet JP. Mouse plasma glutathione peroxidase: cDNA sequence analysis and renal proximal tubular expression and secretion. J Biol Chem. 1994;269:27066–27073. - PubMed
-
- Maser RL, Calvet JP. Reduced expression of antioxidant and detoxicant enzyme genes in polycystic kidney disease (PKD) J Am Soc Nephrol. 1996;7:1617. abstract.
-
- Maser RL, Vassmer D, Magenheimer BS, Calvet JP. Oxidant stress and reduced antioxidant enzyme protection in polycystic kidney disease. J Am Soc Nephrol. 2002;13:991–999. - PubMed
-
- Iakoubova OA, Pacella LA, Her H, Beier DR. LTW4 protein on mouse chromosome 1 is a member of a family of antioxidant proteins. Genomics. 1997;42:474–478. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
