Optimisation of chemical protein cleavage for erythropoietin semi-synthesis using native chemical ligation
- PMID: 18931805
- PMCID: PMC2898651
- DOI: 10.1039/b811501j
Optimisation of chemical protein cleavage for erythropoietin semi-synthesis using native chemical ligation
Abstract
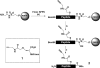
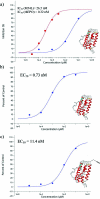
Selective protein cleavage at methionine residues is a useful method for the production of bacterially derived protein fragments containing an N-terminal cysteine residue required for native chemical ligation. Here we describe an optimised procedure for cyanogen bromide-mediated protein cleavage, and ligation of the resulting fragments to afford biologically active proteins.
Figures

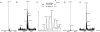
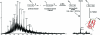
Similar articles
-
Cyanogen bromide cleavage generates fragments suitable for expressed protein and glycoprotein ligation.J Am Chem Soc. 2004 Aug 11;126(31):9530-1. doi: 10.1021/ja047855m. J Am Chem Soc. 2004. PMID: 15291543
-
Cyanogen bromide cleaves Fc fragments of pooled human IgG at both methionine and tryptophan residues.Mol Immunol. 1985 Dec;22(12):1317-22. doi: 10.1016/0161-5890(85)90052-5. Mol Immunol. 1985. PMID: 3831770
-
Towards biomolecular assembly employing extended native chemical ligation in combination with thioester synthesis using an N-->S acyl shift.Biopolymers. 2010;94(4):495-503. doi: 10.1002/bip.21473. Biopolymers. 2010. PMID: 20593460
-
Recent Progress in the Synthesis of Homogeneous Erythropoietin (EPO) Glycoforms.Chembiochem. 2020 Dec 1;21(23):3301-3312. doi: 10.1002/cbic.202000347. Epub 2020 Jul 30. Chembiochem. 2020. PMID: 33210450 Review.
-
Chemical protein synthesis: Inventing synthetic methods to decipher how proteins work.Bioorg Med Chem. 2017 Sep 15;25(18):4926-4937. doi: 10.1016/j.bmc.2017.06.020. Epub 2017 Jun 15. Bioorg Med Chem. 2017. PMID: 28687227 Review.
Cited by
-
Chemoenzymatic Semisynthesis of Proteins.Chem Rev. 2020 Mar 25;120(6):3051-3126. doi: 10.1021/acs.chemrev.9b00450. Epub 2019 Nov 27. Chem Rev. 2020. PMID: 31774265 Free PMC article. Review.
-
At last: erythropoietin as a single glycoform.Angew Chem Int Ed Engl. 2012 Nov 12;51(46):11576-84. doi: 10.1002/anie.201206090. Epub 2012 Sep 25. Angew Chem Int Ed Engl. 2012. PMID: 23012228 Free PMC article. No abstract available.
-
The winding pathway to erythropoietin along the chemistry-biology frontier: a success at last.Angew Chem Int Ed Engl. 2013 Jul 22;52(30):7646-65. doi: 10.1002/anie.201301666. Epub 2013 Jun 17. Angew Chem Int Ed Engl. 2013. PMID: 23775885 Free PMC article. Review.
References
-
- Lemieux G. A., Bertozzi C. R. Trends Biotechnol. 1998;16(12):506. - PubMed
- Kiick K. L. S. E., Tirrell D. A., Bertozzi C. R. Proc. Natl. Acad. Sci. U. S. A. 2002;99(1):19. - PMC - PubMed
- Wang L., Schultz P. G. Angew. Chem., Int. Ed. 2005;44:34.
- Prescher J. A., Bertozzi C. R. Nat. Chem. Biol. 2005;1(1):13. - PubMed
- Lin P.-C., Ueng S.-H., Tseng M.-C., Ko J.-L., Huang K.-T., Yu S.-C., Adak A. K., Chen Y.-J., Lin C. C. Angew. Chem., Int. Ed. 2006;45(26):4286. - PubMed
- van Kasteren S. I., Kramer H. B., Jensen H. H., Campbell S. J., Kirkpatrick J., Oldham N. J., Anthony D. C., Davis B. G. Nature. 2007;446(7139):1105. - PubMed
- Heal W. P., Wickramasinghe S. R., Leatherbarrow R. J., Tate E. W. Org. Biomol. Chem. 2008;6(13):2308. - PubMed
-
- Noren C. J., Wang J. M., Perler F. B. Angew. Chem., Int. Ed. 2000;39(3):451. - PubMed
- Yeo D. S. Y., Srinivasan R., Chen G. Y. J., Yao S. Q. Chem.–Eur. J. 2004;10(19):4664. - PubMed
- Camarero J. A., Kwon Y., Coleman M. A. J. Am. Chem. Soc. 2004;126(45):14730. - PubMed
- Pellois J. P., Muir T. W. Curr. Opin. Chem. Biol. 2006;10(5):487. - PubMed
- Pickin K. A., Chaudhury S., Dancy B. C. R., Gray J. J., Cole P. A. J. Am. Chem. Soc. 2008;130(17):5667. - PMC - PubMed
- McGinty R. K., Kim J., Chatterjee C., Roeder R. G., Muir T. W. Nature. 2008;453(7196):812. - PMC - PubMed
-
- Macmillan D., Arham L., J. Am. Chem. Soc., 2004, 12631, 9530, . A similar method, operating without the aid of a polyhistidine tag, was also published by Lu and co-workers: Pal G., Santamaria F., Kossiakoff A. A., Lu W., Protein Expression Purif., 2003, 29, 185
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

