Gene expression profiles in zebrafish brain after acute exposure to domoic acid at symptomatic and asymptomatic doses
- PMID: 18936300
- PMCID: PMC2735419
- DOI: 10.1093/toxsci/kfn207
Gene expression profiles in zebrafish brain after acute exposure to domoic acid at symptomatic and asymptomatic doses
Abstract
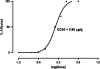
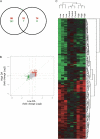
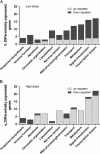
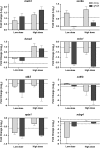
Domoic acid (DA) is a neuroexcitatory amino acid that is naturally produced by some marine diatom species of the genus Pseudo-nitzschia. Ingestion of DA-contaminated seafood by humans results in a severe neurotoxic disease known as amnesic shellfish poisoning (ASP). Clinical signs of ASP include seizures and neuronal damage from activation of ionotropic glutamate receptors. However, the impacts of DA exposure at levels below those known to induce outward signs of neurobehavioral exicitotoxicity have not been well characterized. To further understand the mechanisms of neurotoxic injury associated with DA exposure, we examined the transcriptome of whole brains from zebrafish (Danio rerio) receiving intracoelomic (IC) injection of DA at both symptomatic and asymptomatic doses. A majority of zebrafish exposed to high-dose DA (1.2 microg DA/g) exhibited clinical signs of neuroexcitotoxicity (EC(50) of 0.86 microg DA/g) within 5-20 min of IC injection. All zebrafish receiving low-dose DA (0.47 microg DA/g) or vehicle only maintained normal behavior. Microarray analysis of symptomatic and asymptomatic exposures collectively yielded 306 differentially expressed genes (1.5-fold, p </= 0.05) predominately represented by signal transduction, ion transport, and transcription factor functional categories. Transcriptional profiles were suggestive of neuronal apoptosis following an overwhelming of protective adaptive pathways. Further, potential molecular biomarkers of neuropathic injury, including the zebrafish homolog of human NDRG4, were identified and may be relevant to DA exposure levels below that causing neurobehavioral injury. In general, DA-modulated gene expression was consistent with other model species thereby validating zebrafish as an appropriate vertebrate model to study mechanisms of DA neurotoxicity. These data provide a basis for identifying pathways of DA-induced injury as well as biomarkers of asymptomatic and symptomatic DA exposure levels.
Figures

Similar articles
-
Symptomatic and asymptomatic domoic acid exposure in zebrafish (Danio rerio) revealed distinct non-overlapping gene expression patterns in the brain.Aquat Toxicol. 2022 Nov;252:106310. doi: 10.1016/j.aquatox.2022.106310. Epub 2022 Sep 23. Aquat Toxicol. 2022. PMID: 36198224 Free PMC article.
-
Uptake, tissue distribution and excretion of domoic acid after oral exposure in coho salmon (Oncorhynchus kisutch).Aquat Toxicol. 2007 Mar 10;81(3):266-74. doi: 10.1016/j.aquatox.2006.12.009. Epub 2007 Jan 3. Aquat Toxicol. 2007. PMID: 17250904
-
Domoic acid induces developmental stage-specific effects on microglia in zebrafish.Harmful Algae. 2025 Jun;146:102862. doi: 10.1016/j.hal.2025.102862. Epub 2025 Apr 17. Harmful Algae. 2025. PMID: 40409865
-
Domoic acid and human exposure risks: a review.Toxicon. 2010 Aug 15;56(2):218-30. doi: 10.1016/j.toxicon.2009.05.034. Epub 2009 Jun 6. Toxicon. 2010. PMID: 19505488 Review.
-
Domoic acid: neurobehavioral consequences of exposure to a prevalent marine biotoxin.Neurotoxicol Teratol. 2010 Mar-Apr;32(2):132-41. doi: 10.1016/j.ntt.2009.09.005. Epub 2009 Sep 30. Neurotoxicol Teratol. 2010. PMID: 19799996 Free PMC article. Review.
Cited by
-
In vivo examination of pathogenicity and virulence in environmentally isolated Vibrio vulnificus.Microbiologyopen. 2024 Aug;13(4):e1427. doi: 10.1002/mbo3.1427. Microbiologyopen. 2024. PMID: 39041461 Free PMC article.
-
Prolonged, Low-Level Exposure to the Marine Toxin, Domoic Acid, and Measures of Neurotoxicity in Nonhuman Primates.Environ Health Perspect. 2022 Sep;130(9):97003. doi: 10.1289/EHP10923. Epub 2022 Sep 14. Environ Health Perspect. 2022. PMID: 36102641 Free PMC article.
-
Public health risks associated with chronic, low-level domoic acid exposure: A review of the evidence.Pharmacol Ther. 2021 Nov;227:107865. doi: 10.1016/j.pharmthera.2021.107865. Epub 2021 Apr 28. Pharmacol Ther. 2021. PMID: 33930455 Free PMC article. Review.
-
Domoic acid as a developmental neurotoxin.Neurotoxicology. 2010 Sep;31(5):409-23. doi: 10.1016/j.neuro.2010.05.003. Epub 2010 May 13. Neurotoxicology. 2010. PMID: 20471419 Free PMC article. Review.
-
A novel antibody-based biomarker for chronic algal toxin exposure and sub-acute neurotoxicity.PLoS One. 2012;7(5):e36213. doi: 10.1371/journal.pone.0036213. Epub 2012 May 2. PLoS One. 2012. PMID: 22567140 Free PMC article.
References
-
- Ananth C, Gopalakrishnakone P, Kaur C. Induction of inducible nitric oxide synthase expression in activated microglia following domoic acid (DA)-induced neurotoxicity in the rat hippocampus. Neurosci. Lett. 2003;338:49–52. - PubMed
-
- Ananth C, Thameem Dheen S, Gopalakrishnakone P, Kaur C. Domoic acid-induced neuronal damage in the rat hippocampus: Changes in apoptosis related genes (bcl-2, bax, caspase-3) and microglial response. J. Neurosci. Res. 2001;66:177–190. - PubMed
-
- Ball MJ. Neuronal loss, neurofibrillary tangles and granulovacuolar degeneration in the hippocampus with ageing and dementia. A quantitative study. Acta Neuropathol. 1977;37:111–118. - PubMed
-
- Bates SS, Garrison DL, Horner RA. Bloom dynamics and physiology of domoic-acid-producing Pseudo-nitzschia species. In: Anderson DM, Hallegraeff GM, Cembella AD, editors. The Physiological Ecology of Harmful Algal Blooms-NATO Advanced Study Institute Series. Heidelberg, Germany: Springer-Verlag; 1998. pp. 267–292.
-
- Behrens A, Sibilia M, Wagner EF. Amino-terminal phosphorylation of c-Jun regulates stress-induced apoptosis and cellular proliferation. Nat. Genet. 1999;21:326–329. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

