Human Discs Large is a new negative regulator of human immunodeficiency virus-1 infectivity
- PMID: 18946087
- PMCID: PMC2613124
- DOI: 10.1091/mbc.e08-02-0189
Human Discs Large is a new negative regulator of human immunodeficiency virus-1 infectivity
Abstract
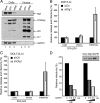
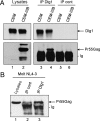
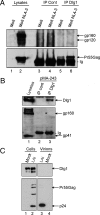
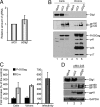
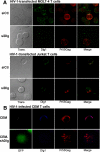
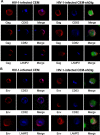
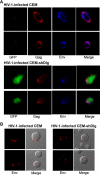
Human immunodeficiency virus (HIV)-1 replication is positively or negatively regulated through multiple interactions with host cell proteins. We report here that human Discs Large (Dlg1), a scaffold protein recruited beneath the plasma membrane and involved in the assembly of multiprotein complexes, restricts HIV-1 infectivity. The endogenous Dlg1 and HIV-1 Gag polyprotein spontaneously interact in HIV-1-chronically infected T cells. Depleting endogenous Dlg1 in either adherent cells or T cells does not affect Gag maturation, production, or release, but it enhances the infectivity of progeny viruses five- to sixfold. Conversely, overexpression of Dlg1 reduces virus infectivity by approximately 80%. Higher virus infectivity upon Dlg1 depletion correlates with increased Env content in cells and virions, whereas the amount of virus-associated Gag or genomic RNA remains identical. Dlg1 knockdown is also associated with the redistribution and colocalization of Gag and Env toward CD63 and CD82 positive vesicle-like structures, including structures that seem to still be connected to the plasma membrane. This study identifies both a new negative regulator that targets the very late steps of the HIV-1 life cycle, and an assembly pathway that optimizes HIV-1 infectivity.
Figures

Similar articles
-
A role for CD81 on the late steps of HIV-1 replication in a chronically infected T cell line.Retrovirology. 2009 Mar 11;6:28. doi: 10.1186/1742-4690-6-28. Retrovirology. 2009. PMID: 19284574 Free PMC article.
-
The scaffolding protein Dlg1 is a negative regulator of cell-free virus infectivity but not of cell-to-cell HIV-1 transmission in T cells.PLoS One. 2012;7(1):e30130. doi: 10.1371/journal.pone.0030130. Epub 2012 Jan 17. PLoS One. 2012. PMID: 22272285 Free PMC article.
-
Modulation of human immunodeficiency virus type 1 infectivity through incorporation of tetraspanin proteins.J Virol. 2008 Jan;82(2):1021-33. doi: 10.1128/JVI.01044-07. Epub 2007 Nov 7. J Virol. 2008. PMID: 17989173 Free PMC article.
-
Roles of the interactions between Env and Gag proteins in the HIV-1 replication cycle.Microbiol Immunol. 2008 May;52(5):287-95. doi: 10.1111/j.1348-0421.2008.00008.x. Microbiol Immunol. 2008. PMID: 18557900 Review.
-
SERINC as a Restriction Factor to Inhibit Viral Infectivity and the Interaction with HIV.J Immunol Res. 2017;2017:1548905. doi: 10.1155/2017/1548905. Epub 2017 Nov 22. J Immunol Res. 2017. PMID: 29359168 Free PMC article. Review.
Cited by
-
How HIV-1 Gag Manipulates Its Host Cell Proteins: A Focus on Interactors of the Nucleocapsid Domain.Viruses. 2020 Aug 13;12(8):888. doi: 10.3390/v12080888. Viruses. 2020. PMID: 32823718 Free PMC article. Review.
-
Nature, nurture and HIV: The effect of producer cell on viral physiology.Virology. 2013 Sep 1;443(2):208-13. doi: 10.1016/j.virol.2013.05.023. Epub 2013 Jun 5. Virology. 2013. PMID: 23747196 Free PMC article. Review.
-
Multiple Inhibitory Factors Act in the Late Phase of HIV-1 Replication: a Systematic Review of the Literature.Microbiol Mol Biol Rev. 2018 Jan 10;82(1):e00051-17. doi: 10.1128/MMBR.00051-17. Print 2018 Mar. Microbiol Mol Biol Rev. 2018. PMID: 29321222 Free PMC article.
-
A role for CD81 on the late steps of HIV-1 replication in a chronically infected T cell line.Retrovirology. 2009 Mar 11;6:28. doi: 10.1186/1742-4690-6-28. Retrovirology. 2009. PMID: 19284574 Free PMC article.
-
PDZD8 is a novel Gag-interacting factor that promotes retroviral infection.J Virol. 2010 Sep;84(17):8990-5. doi: 10.1128/JVI.00843-10. Epub 2010 Jun 23. J Virol. 2010. PMID: 20573829 Free PMC article.
References
-
- Batonick M., Favre M., Boge M., Spearman P., Honing S., Thali M. Interaction of HIV-1 Gag with the clathrin-associated adaptor AP-2. Virology. 2005;342:190–200. - PubMed
-
- Blot V., Delamarre L., Perugi F., Pham D., Benichou S., Benarous R., Hanada T., Chishti A. H., Dokhelar M. C., Pique C. Human Dlg protein binds to the envelope glycoproteins of human T-cell leukemia virus type 1 and regulates envelope mediated cell-cell fusion in T lymphocytes. J. Cell Sci. 2004;117:3983–3993. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

