Platelet-derived growth factor receptor beta signaling is required for efficient epicardial cell migration and development of two distinct coronary vascular smooth muscle cell populations
- PMID: 18948621
- PMCID: PMC2757035
- DOI: 10.1161/CIRCRESAHA.108.176768
Platelet-derived growth factor receptor beta signaling is required for efficient epicardial cell migration and development of two distinct coronary vascular smooth muscle cell populations
Abstract
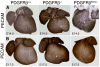
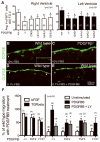
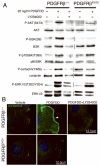
The epicardium plays an essential role in coronary artery formation and myocardial development, but signals controlling the development and differentiation of this tissue are not well understood. To investigate the role of platelet-derived growth factor receptor (PDGFR)beta in development of epicardial-derived vascular smooth muscle cells (VSMCs), we examined PDGFRbeta(-/-) and PDGFRbeta epicardial mutant hearts. We found that PDGFRbeta(-/-) hearts failed to form dominant coronary vessels on the ventral heart surface, had a thinned myocardium, and completely lacked coronary VSMCs (cVSMCs). This constellation of defects was consistent with a primary defect in the epicardium. To verify that these defects were specific to epicardial derivatives, we generated mice with an epicardial deletion of PDGFRbeta that resulted in reduced cVSMCs distal to the aorta. The regional absence of cVSMCs suggested that cVSMCs could arise from 2 sources, epicardial and nonepicardial, and that both were dependent on PDGFRbeta. In the absence of PDGFRbeta signaling, epicardial cells adopted an irregular actin cytoskeleton, leading to aberrant migration of epicardial cells into the myocardium in vivo. In addition, PDGF receptor stimulation promoted epicardial cell migration, and PDGFRbeta-driven phosphoinositide 3'-kinase signaling was critical for this process. Our data demonstrate that PDGFRbeta is required for the formation of 2 distinct cVSMC populations and that loss of PDGFRbeta-PI3K signaling disrupts epicardial cell migration.
Figures




References
-
- Majesky MW. Developmental basis of vascular smooth muscle diversity. Arterioscler Thromb Vasc Biol. 2007;27:1248–1258. - PubMed
-
- Gittenberger-de Groot AC, Vrancken Peeters MP, Mentink MM, Gourdie RG, Poelmann RE. Epicardium-derived cells contribute a novel population to the myocardial wall and the atrioventricular cushions. Circ Res. 1998;82:1043–1052. - PubMed
-
- Mikawa T, Gourdie RG. Pericardial mesoderm generates a population of coronary smooth muscle cells migrating into the heart along with ingrowth of the epicardial organ. Dev Biol. 1996;174:221–232. - PubMed
-
- Hellstrom M, Kalen M, Lindahl P, Abramsson A, Betsholtz C. Role of PDGF-B and PDGFR-beta in recruitment of vascular smooth muscle cells and pericytes during embryonic blood vessel formation in the mouse. Development. 1999;126:3047–3055. - PubMed
-
- Leveen P, Pekny M, Gebre-Medhin S, Swolin B, Larsson E, Betsholtz C. Mice deficient for PDGF B show renal, cardiovascular, and hematological abnormalities. Genes Dev. 1994;8:1875–1887. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

