An expanded biological repertoire for Ins(3,4,5,6)P4 through its modulation of ClC-3 function
- PMID: 18951024
- PMCID: PMC2631652
- DOI: 10.1016/j.cub.2008.08.073
An expanded biological repertoire for Ins(3,4,5,6)P4 through its modulation of ClC-3 function
Abstract
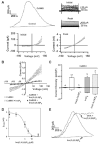
Ins(3,4,5,6)P(4) inhibits plasma membrane Cl(-) flux in secretory epithelia [1]. However, in most other mammalian cells, receptor-dependent elevation of Ins(3,4,5,6)P(4) levels is an "orphan" response that lacks biological significance [2]. We set out to identify Cl(-) channel(s) and/or transporter(s) that are regulated by Ins(3,4,5,6)P4 in vivo. Several candidates [3-5] were excluded through biophysical criteria, electrophysiological analysis, and confocal immunofluorescence microscopy. Then, we heterologously expressed ClC-3 in the plasma membrane of HEK293-tsA201 cells; whole-cell patch-clamp analysis showed Ins(3,4,5,6)P4 to inhibit Cl(-) conductance through ClC-3. Next, we heterologously expressed ClC-3 in the early endosomal compartment of BHK cells; by fluorescence ratio imaging of endocytosed FITC-transferrin, we recorded intra-endosomal pH, an in situ biosensor for Cl(-) flux across endosomal membranes [6]. A cell-permeant, bioactivatable Ins(3,4,5,6)P4 analog elevated endosomal pH from 6.1 to 6.6, reflecting inhibition of ClC-3. Finally, Ins(3,4,5,6)P(4) inhibited endogenous ClC-3 conductance in postsynaptic membranes of neonatal hippocampal neurones. Among other ClC-3 functions that could be regulated by Ins(3,4,5,6)P4 are tumor cell migration [7], apoptosis [8], and inflammatory responses [9]. Ins(3,4,5,6)P4 is a ubiquitous cellular signal with diverse biological actions.
Figures


Comment in
-
Anion channels: regulation of ClC-3 by an orphan second messenger.Curr Biol. 2008 Nov 25;18(22):R1061-4. doi: 10.1016/j.cub.2008.09.023. Curr Biol. 2008. PMID: 19036336
References
-
- Yang L, Reece J, Gabriel SE, Shears SB. Apical localization of ITPK1 enhances its ability to be a modifier gene product in a murine tracheal cell model of cystic fibrosis. J Cell Sci. 2006;119:1320–1328. - PubMed
-
- Menniti FS, Oliver KG, Putney JW, Jr, Shears SB. Inositol phosphates and cell signalling: New views of InsP5 and InsP6. Trends Biochem Sci. 1993;18:53–56. - PubMed
-
- Eggermont J. Calcium-activated chloride channels: (un)known, (un)loved? Proc Am Thorac Soc. 2004;1:22–27. - PubMed
-
- Kunzelmann K, Milenkovic VM, Spitzner M, Soria RB, Schreiber R. Calcium-dependent chloride conductance in epithelia: is there a contribution by Bestrophin? Pflugers Arch. 2007;454:879–889. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

