Agonist-dependent phosphorylation of the formyl peptide receptor is regulated by the membrane proximal region of the cytoplasmic tail
- PMID: 18952127
- PMCID: PMC2709459
- DOI: 10.1016/j.bbamcr.2008.09.011
Agonist-dependent phosphorylation of the formyl peptide receptor is regulated by the membrane proximal region of the cytoplasmic tail
Abstract
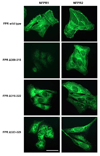
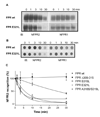
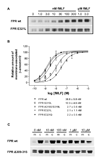
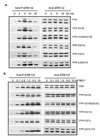
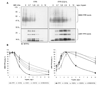
Formyl peptide receptor (FPR) is a chemoattractant G protein-coupled receptor (GPCR) involved in the innate immune response against bacteria. Receptor activation is terminated by receptor phosphorylation of two serine- and threonine-rich regions located in the distal half of the cytoplasmic tail. In this study we show that introduction of an amino acid with a bulky side chain (leucine or glutamine) adjacent to a single leucine, L320, in the membrane-proximal half of the cytoplasmic tail, significantly enhanced receptor phosphorylation, beta-arrestin1/2 translocation, and receptor endocytosis, without affecting G(i)-mediated ERK1/2 activation and release of intracellular calcium. In addition, the point mutations resulted in diminished susceptibility to trypsin, suggesting a conformation different from that of wild type FPR. Alignment of the FPR sequence with the rhodopsin sequence showed that L320 resides immediately C-terminal of an amphipathic region that in rhodopsin forms helix 8. Deletion of seven amino acids (Delta309-315) from the predicted helix 8 of FPR (G307-S319) caused reduced cell signaling as well as defects in receptor phosphorylation, beta-arrestin1/2 translocation and endocytosis. Thus, the amino acid content in the N-terminal half of the cytoplasmic tail influences the structure and desensitization of FPR.
Figures








References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

