Role of dendritic cells in enhancement of herpes simplex virus type 1 latency and reactivation in vaccinated mice
- PMID: 18971304
- PMCID: PMC2593179
- DOI: 10.1128/CVI.00318-08
Role of dendritic cells in enhancement of herpes simplex virus type 1 latency and reactivation in vaccinated mice
Abstract
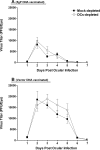
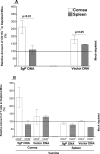
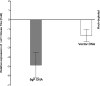
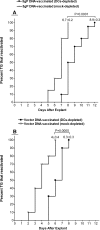
Ocular infection with herpes simplex virus type 1 (HSV-1) frequently leads to recurrent infection, which is a major cause of corneal scarring. Thus, the prevention of the establishment of latency should be a primary goal of vaccination against HSV-1. To this end, we have examined the contribution of dendritic cells (DCs) to the efficacy of a vaccine against ocular HSV-1 infection. Transgenic mice (expressing a CD11c-diphtheria toxin receptor-green fluorescent protein construct) with a BALB/c background were immunized with a vaccine consisting of DNA that encodes five HSV-1 glycoproteins or were immunized with vector control DNA. The vaccinated mice were then depleted of their DCs through the injection of diphtheria toxin before and after ocular challenge with HSV-1. Analyses of HSV-1 replication in the eye, blepharitis, corneal scarring, and the survival of the infected mice upon primary infection indicated that DC depletion neither promoted nor compromised the efficacy of the vaccine. In contrast, DC depletion was associated with an approximately fivefold reduction in the level of latent virus in the trigeminal ganglia (TGs) of latently infected mice, as well as a significant reduction in the reactivation rate of latent virus. The possibility that DCs enhance the latency of HSV-1 in the TGs of ocularly infected mice suggests for the first time that DCs, rather than acting as "immune saviors," can exacerbate disease and compromise vaccine efficacy by enhancing viral latency and reactivation.
Figures

Similar articles
-
An M2 Rather than a TH2 Response Contributes to Better Protection against Latency Reactivation following Ocular Infection of Naive Mice with a Recombinant Herpes Simplex Virus 1 Expressing Murine Interleukin-4.J Virol. 2018 Apr 27;92(10):e00051-18. doi: 10.1128/JVI.00051-18. Print 2018 May 15. J Virol. 2018. PMID: 29491152 Free PMC article.
-
Role of CD8+ T cells and lymphoid dendritic cells in protection from ocular herpes simplex virus 1 challenge in immunized mice.J Virol. 2014 Jul;88(14):8016-27. doi: 10.1128/JVI.00913-14. Epub 2014 May 7. J Virol. 2014. PMID: 24807710 Free PMC article.
-
Lymphoid-related CD11c+ CD8alpha+ dendritic cells are involved in enhancing herpes simplex virus type 1 latency.J Virol. 2008 Oct;82(20):9870-9. doi: 10.1128/JVI.00566-08. Epub 2008 Jul 30. J Virol. 2008. PMID: 18667491 Free PMC article.
-
Control of HSV-1 latency in human trigeminal ganglia--current overview.J Neurovirol. 2011 Dec;17(6):518-27. doi: 10.1007/s13365-011-0063-0. Epub 2011 Dec 3. J Neurovirol. 2011. PMID: 22139603 Review.
-
CD8+ T cells patrol HSV-1-infected trigeminal ganglia and prevent viral reactivation.J Neurovirol. 2011 Dec;17(6):528-34. doi: 10.1007/s13365-011-0062-1. Epub 2011 Dec 8. J Neurovirol. 2011. PMID: 22161682 Review.
Cited by
-
Interrelationship of Primary Virus Replication, Level of Latency, and Time to Reactivation in the Trigeminal Ganglia of Latently Infected Mice.J Virol. 2016 Sep 29;90(20):9533-42. doi: 10.1128/JVI.01373-16. Print 2016 Oct 15. J Virol. 2016. PMID: 27512072 Free PMC article.
-
Essential role of M1 macrophages in blocking cytokine storm and pathology associated with murine HSV-1 infection.PLoS Pathog. 2021 Oct 15;17(10):e1009999. doi: 10.1371/journal.ppat.1009999. eCollection 2021 Oct. PLoS Pathog. 2021. PMID: 34653236 Free PMC article.
-
Binding of herpesvirus entry mediator (HVEM) and HSV-1 gD affect reactivation but not latency levels.PLoS Pathog. 2023 Sep 22;19(9):e1011693. doi: 10.1371/journal.ppat.1011693. eCollection 2023 Sep. PLoS Pathog. 2023. PMID: 37738264 Free PMC article.
-
Herpes Simplex Virus Evasion of Early Host Antiviral Responses.Front Cell Infect Microbiol. 2019 Apr 30;9:127. doi: 10.3389/fcimb.2019.00127. eCollection 2019. Front Cell Infect Microbiol. 2019. PMID: 31114761 Free PMC article. Review.
-
CCR2+ migratory macrophages with M1 status are the early-responders in the cornea of HSV-1 infected mice.PLoS One. 2019 Apr 18;14(4):e0215727. doi: 10.1371/journal.pone.0215727. eCollection 2019. PLoS One. 2019. PMID: 30998796 Free PMC article.
References
-
- Albers, I., H. Kirchner, and I. Domke-Opitz. 1989. Resistance of human blood monocytes to infection with herpes simplex virus. Virology 169:466-469. - PubMed
-
- Albert, M. L., B. Sauter, and N. Bhardwaj. 1998. Dendritic cells acquire antigen from apoptotic cells and induce class I-restricted CTLs. Nature 392:86-89. - PubMed
-
- Banchereau, J., and R. M. Steinman. 1998. Dendritic cells and the control of immunity. Nature 392:245-252. - PubMed
-
- Barber, D. L., E. J. Wherry, D. Masopust, B. Zhu, J. P. Allison, A. H. Sharpe, G. J. Freeman, and R. Ahmed. 2006. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439:682-687. - PubMed
-
- Barron, B. A., L. Gee, W. W. Hauck, N. Kurinij, C. R. Dawson, D. B. Jones, K. R. Wilhelmus, H. E. Kaufman, J. Sugar, R. A. Hyndiuk, et al. 1994. Herpetic eye disease study. A controlled trial of oral acyclovir for herpes simplex stromal keratitis. Ophthalmology 101:1871-1882. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

