Expression of membrane type 1 matrix metalloproteinase (MMP-14) in epithelial ovarian cancer: high level expression in clear cell carcinoma
- PMID: 18976802
- PMCID: PMC2663392
- DOI: 10.1016/j.ygyno.2008.09.025
Expression of membrane type 1 matrix metalloproteinase (MMP-14) in epithelial ovarian cancer: high level expression in clear cell carcinoma
Abstract
Objective: Clear cell carcinomas of the ovary constitute approximately 5% of all ovarian neoplasms and have a distinct gene expression profile relative to other ovarian carcinoma histotypes. Tumors often present as an early stage large pelvic mass with a high degree of recurrence and frequent early metastasis. Matrix metalloproteinases (MMPs) play a role in intraperitoneal metastasis through breakdown of cell-cell and cell-matrix barriers, enabling anchoring of secondary lesions and promoting proliferation in a geometrically constrained matrix environment. The objective of this study was to evaluate MMP expression in ovarian clear cell carcinoma.
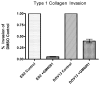
Methods: Immunohistochemistry was used to evaluate expression of membrane type 1 MMP (MMP-14), MMP-2 and MMP-9 in a panel of ovarian tumors. Western blotting and gelatin zymography were used to examine MMP-14 expression and activity in the clear cell carcinoma cell line ES2. The ability of ES2 cells to invade and proliferate within three-dimensional collagen gels was evaluated.
Results: High level expression of MMP-14 and MMP-2 were observed in ovarian clear cell carcinoma relative to other histotypes (94-95% strong positive). MMP-14 was expressed and active in cultured ES2 cells. ES2 cells also exhibited MMP-dependent invasion of and proliferation within three-dimensional collagen gels.
Conclusions: The high level expression of MMP-14 together with in vitro functional analyses suggest that MMP-14 may contribute to both the proliferative capacity and the enhanced parenchymal metastasis of ovarian clear cell carcinoma.
Figures





References
-
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, Thun MJ. Cancer statistics, 2008. CA, A Cancer Journal for Clinicians. 2008;58:71–96. - PubMed
-
- Schwartz DR, Kardia SLR, Shedded KA, et al. Gene expression in ovarian cancer reflects both morphology and biological behavior, distinguishing clear cell from other poor-prognosis ovarian carcinomas. Cancer Research. 2002;62:4722–29. - PubMed
-
- Scully ER, Young RH, Clement PB. Atlas of tumor pathology, third series, fascicle 23. Washington DC: Armed Forces Institute of Pathology; 1998. Tumors of the ovary, maldeveloped gonads, fallopian tube and broad ligament.
-
- DeFrias DVS, Okonkwo AM, Keh PC, Nayar R. Cytopathology of the ovary. In: Stack MS, Fishman DA, editors. Cancer Treatment and Research. Vol. 107. 2002. pp. 185–211. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

