The Anaplasma phagocytophilum PleC histidine kinase and PleD diguanylate cyclase two-component system and role of cyclic Di-GMP in host cell infection
- PMID: 18978058
- PMCID: PMC2632110
- DOI: 10.1128/JB.01218-08
The Anaplasma phagocytophilum PleC histidine kinase and PleD diguanylate cyclase two-component system and role of cyclic Di-GMP in host cell infection
Abstract
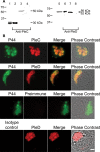
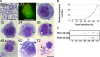
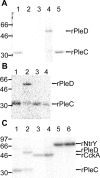
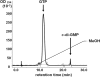
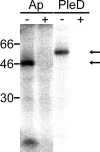
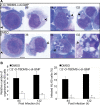
Anaplasma phagocytophilum, the etiologic agent of human granulocytic anaplasmosis (HGA), has genes predicted to encode three sensor kinases, one of which is annotated PleC, and three response regulators, one of which is PleD. Prior to this study, the roles of PleC and PleD in the obligatory intracellular parasitism of A. phagocytophilum and their biochemical activities were unknown. The present study illustrates the relevance of these factors by demonstrating that both pleC and pleD were expressed in an HGA patient. During A. phagocytophilum development in human promyelocytic HL-60 cells, PleC and PleD were synchronously upregulated at the exponential growth stage and downregulated prior to extracellular release. A recombinant PleC kinase domain (rPleCHKD) has histidine kinase activity; no activity was observed when the conserved site of phosphorylation was replaced with alanine. A recombinant PleD (rPleD) has autokinase activity using phosphorylated rPleCHKD as the phosphoryl donor but not with two other recombinant histidine kinases. rPleCHKD could not serve as the phosphoryl donor for a mutant rPleD (with a conserved aspartic acid, the site of phosphorylation, replaced by alanine) or two other A. phagocytophilum recombinant response regulators. rPleD had diguanylate cyclase activity to generate cyclic (c) di-GMP from GTP in vitro. UV cross-linking of A. phagocytophilum lysate with c-di-[(32)P]GMP detected an approximately 47-kDa endogenous protein, presumably c-di-GMP downstream receptor. A new hydrophobic c-di-GMP derivative, 2'-O-di(tert-butyldimethylsilyl)-c-di-GMP, inhibited A. phagocytophilum infection in HL-60 cells. Our results suggest that the two-component PleC-PleD system is a diguanylate cyclase and that a c-di-GMP-receptor complex regulates A. phagocytophilum intracellular infection.
Figures
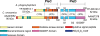

Comment in
-
Cyclic Di-GMP (c-Di-GMP) goes into host cells--c-Di-GMP signaling in the obligate intracellular pathogen Anaplasma phagocytophilum.J Bacteriol. 2009 Feb;191(3):683-6. doi: 10.1128/JB.01593-08. Epub 2008 Dec 1. J Bacteriol. 2009. PMID: 19047357 Free PMC article. No abstract available.
Similar articles
-
Cyclic Di-GMP (c-Di-GMP) goes into host cells--c-Di-GMP signaling in the obligate intracellular pathogen Anaplasma phagocytophilum.J Bacteriol. 2009 Feb;191(3):683-6. doi: 10.1128/JB.01593-08. Epub 2008 Dec 1. J Bacteriol. 2009. PMID: 19047357 Free PMC article. No abstract available.
-
Intra-leukocyte expression of two-component systems in Ehrlichia chaffeensis and Anaplasma phagocytophilum and effects of the histidine kinase inhibitor closantel.Cell Microbiol. 2006 Aug;8(8):1241-52. doi: 10.1111/j.1462-5822.2006.00704.x. Cell Microbiol. 2006. PMID: 16882029
-
Cyclic dimeric GMP signaling regulates intracellular aggregation, sessility, and growth of Ehrlichia chaffeensis.Infect Immun. 2011 Oct;79(10):3905-12. doi: 10.1128/IAI.05320-11. Epub 2011 Jul 25. Infect Immun. 2011. PMID: 21788390 Free PMC article.
-
[Activity of cyclic diguanylate (c-di-GMP) in bacteria and the study of its derivatives].Yao Xue Xue Bao. 2012 Mar;47(3):307-12. Yao Xue Xue Bao. 2012. PMID: 22645753 Review. Chinese.
-
Mechanisms of cyclic-di-GMP signaling in bacteria.Annu Rev Genet. 2006;40:385-407. doi: 10.1146/annurev.genet.40.110405.090423. Annu Rev Genet. 2006. PMID: 16895465 Review.
Cited by
-
A Cyclic di-GMP Network Is Present in Gram-Positive Streptococcus and Gram-Negative Proteus Species.ACS Infect Dis. 2020 Oct 9;6(10):2672-2687. doi: 10.1021/acsinfecdis.0c00314. Epub 2020 Sep 2. ACS Infect Dis. 2020. PMID: 32786278 Free PMC article.
-
Comparative analysis of wolbachia genomes reveals streamlining and divergence of minimalist two-component systems.G3 (Bethesda). 2015 Mar 24;5(5):983-96. doi: 10.1534/g3.115.017137. G3 (Bethesda). 2015. PMID: 25809075 Free PMC article.
-
The cyclic-di-GMP signaling pathway in the Lyme disease spirochete, Borrelia burgdorferi.Front Cell Infect Microbiol. 2014 May 1;4:56. doi: 10.3389/fcimb.2014.00056. eCollection 2014. Front Cell Infect Microbiol. 2014. PMID: 24822172 Free PMC article. Review.
-
Diversity of Cyclic Di-GMP-Binding Proteins and Mechanisms.J Bacteriol. 2016 Jan 1;198(1):32-46. doi: 10.1128/JB.00333-15. J Bacteriol. 2016. PMID: 26055114 Free PMC article. Review.
-
Aptamer selection against cell extracts containing the zoonotic obligate intracellular bacterium, Anaplasma phagocytophilum.Sci Rep. 2024 Jan 30;14(1):2465. doi: 10.1038/s41598-024-52808-8. Sci Rep. 2024. PMID: 38291133 Free PMC article.
References
-
- Amikam, D., and M. Y. Galperin. 2006. PilZ domain is part of the bacterial c-di-GMP binding protein. Bioinformatics 223-6. - PubMed
-
- Ausmees, N., R. Mayer, H. Weinhouse, G. Volman, D. Amikam, M. Benziman, and M. Lindberg. 2001. Genetic data indicate that proteins containing the GGDEF domain possess diguanylate cyclase activity. FEMS Microbiol. Lett. 204163-167. - PubMed
-
- Bakken, J. S., J. S. Dumler, S. M. Chen, M. R. Eckman, L. L. Van Etta, and D. H. Walker. 1994. Human granulocytic ehrlichiosis in the upper Midwest United States. A new species emerging? JAMA 272212-218. - PubMed
-
- Cheng, Z., Y. Kumagai, M. Lin, C. Zhang, and Y. Rikihisa. 2006. Intra-leukocyte expression of two-component systems in Ehrlichia chaffeensis and Anaplasma phagocytophilum and effects of the histidine kinase inhibitor closantel. Cell. Microbiol. 81241-1252. - PubMed
-
- Christen, B., M. Christen, R. Paul, F. Schmid, M. Folcher, P. Jenoe, M. Meuwly, and U. Jenal. 2006. Allosteric control of cyclic di-GMP signaling. J. Biol. Chem. 28132015-32024. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

