Effect of micrometer-scale roughness of the surface of Ti6Al4V pedicle screws in vitro and in vivo
- PMID: 18978418
- PMCID: PMC2663328
- DOI: 10.2106/JBJS.G.00499
Effect of micrometer-scale roughness of the surface of Ti6Al4V pedicle screws in vitro and in vivo
Abstract
Background: Titanium implants that have been grit-blasted and acid-etched to produce a rough microtopography support more bone integration than do smooth-surfaced implants. In vitro studies have suggested that this is due to a stimulatory effect on osteoblasts. It is not known if grit-blasted and acid-etched Ti6Al4V implants also stimulate osteoblasts and increase bone formation clinically. In this study, we examined the effects of micrometer-scale-structured Ti6Al4V surfaces on cell responses in vitro and on tissue responses in vivo.
Methods: Ti6Al4V disks were either machined to produce smooth surfaces with an average roughness (Ra) of 0.2 microm or grit-blasted, resulting in an Ra of 2.0, 3.0, or 3.3 microm. Human osteoblast-like cells were cultured on the disks and on tissue culture polystyrene. The cell number, markers of osteoblast differentiation, and levels of local factors in the conditioned media were determined at confluence. In addition, Ti6Al4V pedicle screws with smooth or rough surfaces were implanted into the L4 and L5 vertebrae of fifteen two-year-old sheep. Osteointegration was evaluated at twelve weeks with histomorphometry and on the basis of removal torque.
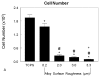
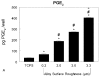
Results: The cell numbers on the Ti6Al4V surfaces were lower than those on the tissue culture polystyrene; the effect was greatest on the roughest surface. The alkaline-phosphatase-specific activity of cell lysates was decreased in a surface-dependent manner, whereas osteocalcin, prostaglandin E(2), transforming growth factor-beta1, and osteoprotegerin levels were higher on the rough surfaces. Bone-implant contact was greater around the rough-surfaced Ti6Al4V screws, and the torque needed to remove the rough screws from the bone was more than twice that required to remove the smooth screws.
Conclusions: Increased micrometer-scale surface roughness increases osteoblast differentiation and local factor production in vitro, which may contribute to increased bone formation and osteointegration in vivo. There was a correlation between in vitro and in vivo observations, indicating that the use of screws with rough surfaces will result in better bone-implant contact and implant stability.
Figures













Similar articles
-
Response of normal female human osteoblasts (NHOst) to 17beta-estradiol is modulated by implant surface morphology.J Biomed Mater Res. 2002 Nov;62(2):204-13. doi: 10.1002/jbm.10290. J Biomed Mater Res. 2002. PMID: 12209940
-
Osteoblast-like cells are sensitive to submicron-scale surface structure.Clin Oral Implants Res. 2006 Jun;17(3):258-64. doi: 10.1111/j.1600-0501.2005.01195.x. Clin Oral Implants Res. 2006. PMID: 16672020
-
Arachidonic acid and prostaglandin E2 influence human osteoblast (MG63) response to titanium surface roughness.J Oral Implantol. 2008;34(6):303-12. doi: 10.1563/1548-1336-34.6.303. J Oral Implantol. 2008. PMID: 19133484
-
Underlying mechanisms at the bone-surface interface during regeneration.J Periodontal Res. 1997 Jan;32(1 Pt 2):166-71. doi: 10.1111/j.1600-0765.1997.tb01399.x. J Periodontal Res. 1997. PMID: 9085228 Review.
-
[The role of surface roughness in promoting osteointegration].Refuat Hapeh Vehashinayim (1993). 2003 Jul;20(3):8-19, 98. Refuat Hapeh Vehashinayim (1993). 2003. PMID: 14515625 Review. Hebrew.
Cited by
-
Effects of coating a titanium alloy with fibronectin on the expression of osteoblast gene markers in the MC3T3 osteoprogenitor cell line.Int J Oral Maxillofac Implants. 2012 Sep-Oct;27(5):1081-90. Int J Oral Maxillofac Implants. 2012. PMID: 23057020 Free PMC article.
-
Regenerative medicine: Are calcium phosphate ceramics 'smart' biomaterials?Nat Rev Rheumatol. 2011 Jan;7(1):8-9. doi: 10.1038/nrrheum.2010.210. Nat Rev Rheumatol. 2011. PMID: 21206482 No abstract available.
-
Integrin alpha2beta1 plays a critical role in osteoblast response to micron-scale surface structure and surface energy of titanium substrates.Proc Natl Acad Sci U S A. 2008 Oct 14;105(41):15767-72. doi: 10.1073/pnas.0805420105. Epub 2008 Oct 8. Proc Natl Acad Sci U S A. 2008. PMID: 18843104 Free PMC article.
-
The role of angiogenesis in implant dentistry part I: Review of titanium alloys, surface characteristics and treatments.Med Oral Patol Oral Cir Bucal. 2016 Jul 1;21(4):e514-525. doi: 10.4317/medoral.21199. Med Oral Patol Oral Cir Bucal. 2016. PMID: 27031073 Free PMC article. Review.
-
The effect of the thread depth on the mechanical properties of the dental implant.J Adv Prosthodont. 2015 Apr;7(2):115-21. doi: 10.4047/jap.2015.7.2.115. Epub 2015 Apr 23. J Adv Prosthodont. 2015. PMID: 25932309 Free PMC article.
References
-
- Boyan BD, Lossdörfer S, Wang L, Zhao G, Lohmann CH, Cochran DL, Schwartz Z. Osteoblasts generate an osteogenic microenvironment when grown on surfaces with rough microtopographies. Eur Cell Mater. 2003;6:22-7. - PubMed
-
- Bagno A, Di Bello C. Surface treatments and roughness properties of Ti-based biomaterials. J Mater Sci Mater Med. 2004;15:935-49. - PubMed
-
- Brunette DM. Spreading and orientation of epithelial cells on grooved substrata. Exp Cell Res. 1986;167:203-17. - PubMed
-
- Thomas KA, Cook SD. An evaluation of variables influencing implant fixation by direct bone apposition. J Biomed Mater Res. 1985;19:875-901. - PubMed
-
- Cochran DL. A comparison of endosseous dental implant surfaces. J Periodontol. 1999;70:1523-39. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

