Brain-specific proteins decline in the cerebrospinal fluid of humans with Huntington disease
- PMID: 18984577
- PMCID: PMC2649809
- DOI: 10.1074/mcp.M800231-MCP200
Brain-specific proteins decline in the cerebrospinal fluid of humans with Huntington disease
Abstract
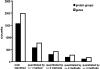
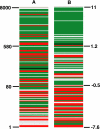
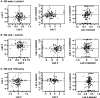
We integrated five sets of proteomics data profiling the constituents of cerebrospinal fluid (CSF) derived from Huntington disease (HD)-affected and -unaffected individuals with genomics data profiling various human and mouse tissues, including the human HD brain. Based on an integrated analysis, we found that brain-specific proteins are 1.8 times more likely to be observed in CSF than in plasma, that brain-specific proteins tend to decrease in HD CSF compared with unaffected CSF, and that 81% of brain-specific proteins have quantitative changes concordant with transcriptional changes identified in different regions of HD brain. The proteins found to increase in HD CSF tend to be liver-associated. These protein changes are consistent with neurodegeneration, microgliosis, and astrocytosis known to occur in HD. We also discuss concordance between laboratories and find that ratios of individual proteins can vary greatly, but the overall trends with respect to brain or liver specificity were consistent. Concordance is highest between the two laboratories observing the largest numbers of proteins.
Figures
References
-
- The Huntington's Disease Collaborative Research Group ( 1993) A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. Cell 72, 971–983 - PubMed
-
- Hodges, A., Strand, A. D., Aragaki, A. K., Kuhn, A., Sengstag, T., Hughes, G., Elliston, L. A., Hartog, C., Goldstein, D. R., Thu, D., Hollingsworth, Z. R., Collin, F., Synek, B., Holmans, P. A., Young, A. B., Wexler, N. S., Delorenzi, M., Kooperberg, C., Augood, S. J., Faull, R. L., Olson, J. M., Jones, L., and Luthi-Carter, R. ( 2006) Regional and cellular gene expression changes in human Huntington's disease brain. Hum. Mol. Genet. 15, 965–977 - PubMed
-
- Chan, E. Y., Luthi-Carter, R., Strand, A., Solano, S. M., Hanson, S. A., DeJohn, M. M., Kooperberg, C., Chase, K. O., DiFiglia, M., Young, A. B., Leavitt, B. R., Cha, J. H., Aronin, N., Hayden, M. R., and Olson, J. M. ( 2002) Increased huntingtin protein length reduces the number of polyglutamine-induced gene expression changes in mouse models of Huntington's disease. Hum. Mol. Genet. 11, 1939–1951 - PubMed
-
- Strand, A. D., Aragaki, A. K., Shaw, D., Bird, T., Holton, J., Turner, C., Tapscott, S. J., Tabrizi, S. J., Schapira, A. H., Kooperberg, C., and Olson, J. M. ( 2005) Gene expression in Huntington's disease skeletal muscle: a potential biomarker. Hum. Mol. Genet. 14, 1863–1876 - PubMed
-
- Luthi-Carter, R., Hanson, S. A., Strand, A. D., Bergstrom, D. A., Chun, W., Peters, N. L., Woods, A. M., Chan, E. Y., Kooperberg, C., Krainc, D., Young, A. B., Tapscott, S. J., and Olson, J. M. ( 2002) Dysregulation of gene expression in the R6/2 model of polyglutamine disease: parallel changes in muscle and brain. Hum. Mol. Genet. 11, 1911–1926 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

