Human immunodeficiency virus type 1 Nef incorporation into virions does not increase infectivity
- PMID: 18987145
- PMCID: PMC2612363
- DOI: 10.1128/JVI.01633-08
Human immunodeficiency virus type 1 Nef incorporation into virions does not increase infectivity
Abstract
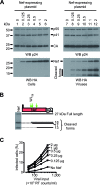
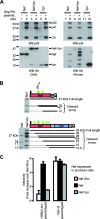
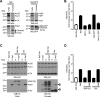
The viral protein Nef contributes to the optimal infectivity of human and simian immunodeficiency viruses. The requirement for Nef during viral biogenesis particles suggests that Nef might play a role in this process. Alternatively, because Nef is incorporated into viruses, it might play a role when progeny virions reach target cells. We challenged these hypotheses by manipulating the amounts of Nef incorporated in viruses while keeping its expression level constant in producer cells. This was achieved by forcing the incorporation of Nef into viral particles by fusing a Vpr sequence to the C-terminal end of Nef. A cleavage site for the viral protease was introduced between Nef and Vpr to allow the release of Nef fragments from the fusion protein during virus maturation. We show that the resulting Nef-CS-Vpr fusion partially retains the ability of Nef to downregulate cell surface CD4 and that high amounts of Nef-CS-Vpr are incorporated into viral particles compared with what is seen for wild-type Nef. The fusion protein is processed during virion maturation and releases Nef fragments similar to those found in viruses produced in the presence of wild-type Nef. Unlike viruses produced in the presence of wild-type Nef, viruses produced in the presence of Nef-CS-Vpr do not have an increase in infectivity and are as poorly infectious as viruses produced in the absence of Nef. These findings demonstrate that the presence of Nef in viral particles is not sufficient to increase human immunodeficiency virus type 1 infectivity and suggest that Nef plays a role during the biogenesis of viral particles.
Figures

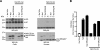
Similar articles
-
Producer-cell modification of human immunodeficiency virus type 1: Nef is a virion protein.J Virol. 1996 Jul;70(7):4283-90. doi: 10.1128/JVI.70.7.4283-4290.1996. J Virol. 1996. PMID: 8676450 Free PMC article.
-
Nef enhances HIV-1 infectivity via association with the virus assembly complex.Virology. 2008 Apr 10;373(2):287-97. doi: 10.1016/j.virol.2007.12.001. Epub 2008 Jan 14. Virology. 2008. PMID: 18191978 Free PMC article.
-
The host-cell restriction factor SERINC5 restricts HIV-1 infectivity without altering the lipid composition and organization of viral particles.J Biol Chem. 2017 Aug 18;292(33):13702-13713. doi: 10.1074/jbc.M117.797332. Epub 2017 Jun 28. J Biol Chem. 2017. PMID: 28659343 Free PMC article.
-
Cell-dependent functional roles of HIV-1 Nef for virus replication (review).Int J Mol Med. 1999 Apr;3(4):427-30. doi: 10.3892/ijmm.3.4.427. Int J Mol Med. 1999. PMID: 10085418 Review.
-
Could Nef and Vpr proteins contribute to disease progression by promoting depletion of bystander cells and prolonged survival of HIV-infected cells?Biochem Biophys Res Commun. 2000 Jan 27;267(3):677-85. doi: 10.1006/bbrc.1999.1708. Biochem Biophys Res Commun. 2000. PMID: 10673351 Review.
Cited by
-
MLV glycosylated-Gag is an infectivity factor that rescues Nef-deficient HIV-1.Proc Natl Acad Sci U S A. 2010 May 18;107(20):9364-9. doi: 10.1073/pnas.1001554107. Epub 2010 May 3. Proc Natl Acad Sci U S A. 2010. PMID: 20439730 Free PMC article.
-
Nef decreases HIV-1 sensitivity to neutralizing antibodies that target the membrane-proximal external region of TMgp41.PLoS Pathog. 2011 Dec;7(12):e1002442. doi: 10.1371/journal.ppat.1002442. Epub 2011 Dec 15. PLoS Pathog. 2011. PMID: 22194689 Free PMC article.
-
Suboptimal provirus expression explains apparent nonrandom cell coinfection with HIV-1.J Virol. 2012 Aug;86(16):8810-20. doi: 10.1128/JVI.00831-12. Epub 2012 Jun 13. J Virol. 2012. PMID: 22696639 Free PMC article.
-
The activity of Nef on HIV-1 infectivity.Front Microbiol. 2014 May 20;5:232. doi: 10.3389/fmicb.2014.00232. eCollection 2014. Front Microbiol. 2014. PMID: 24904546 Free PMC article. Review.
-
The Nef-infectivity enigma: mechanisms of enhanced lentiviral infection.Curr HIV Res. 2011 Oct;9(7):474-89. doi: 10.2174/157016211798842099. Curr HIV Res. 2011. PMID: 22103831 Free PMC article. Review.
References
-
- Ahmad, N., and S. Venkatesan. 1988. Nef protein of HIV-1 is a transcriptional repressor of HIV-1 LTR. Science 2411481-1485. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

