Properties of square-pyramidal alkyl-thiolate Fe(III) complexes, including an analogue of the unmodified form of nitrile hydratase
- PMID: 18989922
- PMCID: PMC2659597
- DOI: 10.1021/ic801704n
Properties of square-pyramidal alkyl-thiolate Fe(III) complexes, including an analogue of the unmodified form of nitrile hydratase
Abstract
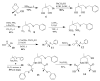
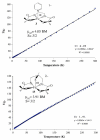
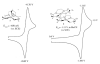
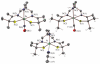
The syntheses and structures of three new coordinatively unsaturated, monomeric, square-pyramidal thiolate-ligated Fe(III) complexes are described, [Fe(III)((tame-N(3))S(2)(Me2))](+) (1), [Fe(III)(Et-N(2)S(2)(Me2))(py)](1-) (3), and [Fe(III)((tame-N(2)S)S(2)(Me2))](2-) (15). The anionic bis-carboxamide, tris-thiolate N(2)S(3) coordination sphere of 15 is potentially similar to that of the yet-to-be characterized unmodified form of NHase. Comparison of the magnetic and reactivity properties of these reveals how anionic charge build up (from cationic 1 to anionic 3 and dianionic 15) and spin-state influence apical ligand affinity. For all of the ligand-field combinations examined, an intermediate S = 3/2 spin state was shown to be favored by a strong N(2)S(2) basal plane ligand field, and this was found to reduce the affinity for apical ligands, even when they are built in. This is in contrast to the post-translationally modified NHase active site, which is low spin and displays a higher affinity for apical ligands. Cationic 1 and its reduced Fe(II) precursor are shown to bind NO and CO, respectively, to afford [Fe(III)((tame-N(3))S(2)(Me))(NO)](+) (18, nu(NuO) = 1865 cm(-1)), an analogue of NO-inactivated NHase, and [Fe(II)((tame-N(3))S(2)(Me))(CO)] (16; nu(CO) stretch (1895 cm(-1)). Anions (N(3)(-), CN(-)) are shown to be unreactive toward 1, 3, and 15 and neutral ligands unreactive toward 3 and 15, even when present in 100-fold excess and at low temperatures. The curtailed reactivity of 15, an analogue of the unmodified form of NHase, and its apical-oxygenated S = 3/2 derivative [Fe(III)((tame-N(2)SO(2))S(2)(Me2))](2-) (20) suggests that regioselective post-translational oxygenation of the basal plane NHase cysteinate sulfurs plays an important role in promoting substrate binding. This is supported by previously reported theoretical (DFT) calculations.
Figures



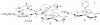




References
-
- Mitra S, Holz RC. J Biol Chem. 2007;282:7397–7404. - PubMed
-
- Noguchi T, Nojiri M, Takei K, Odaka M, Kamiya N. Biochemistry. 2003;42:11642–11650. - PubMed
-
- Kobayashi M, Shimizu S. Nature Biotechnology. 1998;16:733–736. - PubMed
-
- Sugiura Y, Kuwahara J, Nagasawa T, Yamada H. J. Am. Chem. Soc. 1987;109:5848–5850.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

