IL-4 inhibits TGF-beta-induced Foxp3+ T cells and, together with TGF-beta, generates IL-9+ IL-10+ Foxp3(-) effector T cells
- PMID: 18997793
- PMCID: PMC2999006
- DOI: 10.1038/ni.1677
IL-4 inhibits TGF-beta-induced Foxp3+ T cells and, together with TGF-beta, generates IL-9+ IL-10+ Foxp3(-) effector T cells
Erratum in
- Nat Immunol. 2009 May;10(5):551
Abstract
Transcription factor Foxp3 is critical for generating regulatory T cells (T(reg) cells). Transforming growth factor-beta (TGF-beta) induces Foxp3 and suppressive T(reg) cells from naive T cells, whereas interleukin 6 (IL-6) inhibits the generation of inducible T(reg) cells. Here we show that IL-4 blocked the generation of TGF-beta-induced Foxp3(+) T(reg) cells and instead induced a population of T helper cells that produced IL-9 and IL-10. The IL-9(+)IL-10(+) T cells demonstrated no regulatory properties despite producing abundant IL-10. Adoptive transfer of IL-9(+)IL-10(+) T cells into recombination-activating gene 1-deficient mice induced colitis and peripheral neuritis, the severity of which was aggravated if the IL-9(+)IL-10(+) T cells were transferred with CD45RB(hi) CD4(+) effector T cells. Thus IL-9(+)IL-10(+) T cells lack suppressive function and constitute a distinct population of helper-effector T cells that promote tissue inflammation.
Figures

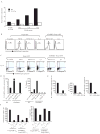
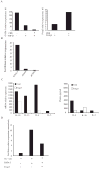


References
-
- Szabo SJea. A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell. 2000:655–69. - PubMed
-
- Zheng WFR. The transcription factor GATA-3 is necessary and sufficient for Th2 cytokine gene expression in CD4 T cells. Cell. 1997:587–96. - PubMed
-
- Ivanov IIea. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell. 2006:1121–33. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 1P01NS38037-04/NS/NINDS NIH HHS/United States
- 2R01NS35685-06/NS/NINDS NIH HHS/United States
- R01 NS045937/NS/NINDS NIH HHS/United States
- R01 NS035685/NS/NINDS NIH HHS/United States
- R01AI073542-01/AI/NIAID NIH HHS/United States
- 2R37NS30843-11/NS/NINDS NIH HHS/United States
- R01 AI073542/AI/NIAID NIH HHS/United States
- R37 NS030843/NS/NINDS NIH HHS/United States
- 2P01A139671-07/PHS HHS/United States
- P01 AI073748/AI/NIAID NIH HHS/United States
- 1R01A144880-03/PHS HHS/United States
- 1R01NS045937-01/NS/NINDS NIH HHS/United States
- R01 NS046414/NS/NINDS NIH HHS/United States
- 1R01NS046414/NS/NINDS NIH HHS/United States
- P01 NS038037/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

