Carbendazim inhibits cancer cell proliferation by suppressing microtubule dynamics
- PMID: 19001156
- PMCID: PMC2682274
- DOI: 10.1124/jpet.108.143537
Carbendazim inhibits cancer cell proliferation by suppressing microtubule dynamics
Abstract
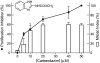
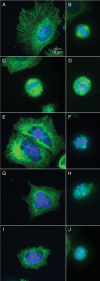
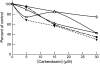
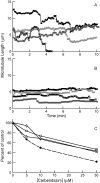
Carbendazim (methyl 2-benzimidazolecarbamate) is widely used as a systemic fungicide in human food production and appears to act on fungal tubulin. However, it also inhibits proliferation of human cancer cells, including drug- and multidrug-resistant and p53-deficient cell lines. Because of its promising preclinical anti-tumor activity, it has undergone phase I clinical trials and is under further clinical development. Although it weakly inhibits polymerization of brain microtubules and induces G(2)/M arrest in tumor cells, its mechanism of action in human cells has not been fully elucidated. We examined its mechanism of action in MCF7 human breast cancer cells and found that it inhibits proliferation (IC(50), 10 microM) and half-maximally arrests mitosis at a similar concentration (8 microM), in concert with suppression of microtubule dynamic instability without appreciable microtubule depolymerization. It induces mitotic spindle abnormalities and reduces the metaphase intercentromere distance of sister chromatids, indicating reduction of tension on kinetochores, thus leading to metaphase arrest. With microtubules assembled in vitro from pure tubulin, carbendazim also suppresses dynamic instability, reducing the dynamicity by 50% at 10 microM, with only minimal (21%) reduction of polymer mass. Carbendazim binds to mammalian tubulin (K(d), 42.8 +/- 4.0 microM). Unlike some benzimidazoles that bind to the colchicine site in tubulin, carbendazim neither competes with colchicine nor competes with vinblastine for binding to brain tubulin. Thus, carbendazim binds to an as yet unidentified site in tubulin and inhibits tumor cell proliferation by suppressing the growing and shortening phases of microtubule dynamic instability, thus inducing mitotic arrest.
Figures
Similar articles
-
Antimitotic antifungal compound benomyl inhibits brain microtubule polymerization and dynamics and cancer cell proliferation at mitosis, by binding to a novel site in tubulin.Biochemistry. 2004 Jun 1;43(21):6645-55. doi: 10.1021/bi036112v. Biochemistry. 2004. PMID: 15157098
-
The primary antimitotic mechanism of action of the synthetic halichondrin E7389 is suppression of microtubule growth.Mol Cancer Ther. 2005 Jul;4(7):1086-95. doi: 10.1158/1535-7163.MCT-04-0345. Mol Cancer Ther. 2005. PMID: 16020666
-
2-Methoxyestradiol suppresses microtubule dynamics and arrests mitosis without depolymerizing microtubules.Mol Cancer Ther. 2006 Sep;5(9):2225-33. doi: 10.1158/1535-7163.MCT-06-0113. Mol Cancer Ther. 2006. PMID: 16985056
-
Microtubules and Cell Division: Potential Pharmacological Targets in Cancer Therapy.Curr Drug Targets. 2023;24(11):889-918. doi: 10.2174/1389450124666230731094837. Curr Drug Targets. 2023. PMID: 37519203 Review.
-
[Inhibitors of microtubule polymerization- new natural compounds as potential anti-cancer drugs].Postepy Hig Med Dosw (Online). 2015 May 4;69:571-85. doi: 10.5604/17322693.1151293. Postepy Hig Med Dosw (Online). 2015. PMID: 25983296 Review. Polish.
Cited by
-
Aneugen Molecular Mechanism Assay: Proof-of-Concept With 27 Reference Chemicals.Toxicol Sci. 2019 Aug 1;170(2):382-393. doi: 10.1093/toxsci/kfz123. Toxicol Sci. 2019. PMID: 31132080 Free PMC article.
-
Benzimidazole and its derivatives as cancer therapeutics: The potential role from traditional to precision medicine.Acta Pharm Sin B. 2023 Feb;13(2):478-497. doi: 10.1016/j.apsb.2022.09.010. Epub 2022 Sep 21. Acta Pharm Sin B. 2023. PMID: 36873180 Free PMC article. Review.
-
Molecular design, synthesis and biological evaluation of novel 1,2,5-trisubstituted benzimidazole derivatives as cytotoxic agents endowed with ABCB1 inhibitory action to overcome multidrug resistance in cancer cells.J Enzyme Inhib Med Chem. 2022 Dec;37(1):2710-2724. doi: 10.1080/14756366.2022.2127700. J Enzyme Inhib Med Chem. 2022. PMID: 36168121 Free PMC article.
-
Neurobehavioral Toxicity Induced by Carbendazim in Rats and the Role of iNOS, Cox-2, and NF-κB Signalling Pathway.Neurochem Res. 2022 Jul;47(7):1956-1971. doi: 10.1007/s11064-022-03581-5. Epub 2022 Mar 21. Neurochem Res. 2022. PMID: 35312909
-
Pre-clinical evaluation of a novel class of anti-cancer agents, the Pyrrolo-1, 5-benzoxazepines.J Cancer. 2016 Dec 4;7(15):2367-2377. doi: 10.7150/jca.16616. eCollection 2016. J Cancer. 2016. PMID: 27994676 Free PMC article. Review.
References
-
- Aneja R, Lopus M, Zhou J, Vangapandu SN, Ghaleb A, Yao J, Nettles JH, Zhou B, Gupta M, Panda D, Chandra R, and Joshi HC (2006) Rational design of the microtubule-targeting anti-breast cancer drug EM015. Cancer Res 66 3782-3791. - PubMed
-
- Bhattacharyya B and Wolff J (1975) The interaction of 1-anilino-8-naphthalene sulfonate with tubulin: a site independent of the colchicine-binding site. Arch Biochem Biophys 167 264-269. - PubMed
-
- Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72 248-254. - PubMed
-
- Brand L and Gohlke JR (1972) Fluorescence probes for structure. Annu Rev Biochem 41 843-868. - PubMed
-
- Can A and Albertini DF (1997) M-phase specific centrosome-microtubule alterations induced by the fungicide MBC in human granulosa cells. Mutat Res 373 139-151. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

