Targeted delivery of PSC-RANTES for HIV-1 prevention using biodegradable nanoparticles
- PMID: 19002569
- PMCID: PMC4243299
- DOI: 10.1007/s11095-008-9765-2
Targeted delivery of PSC-RANTES for HIV-1 prevention using biodegradable nanoparticles
Abstract
Purpose: Nanoparticles formulated from the biodegradable co-polymer poly(lactic-co-glycolic acid) (PLGA), were investigated as a drug delivery system to enhance tissue uptake, permeation, and targeting for PSC-RANTES anti-HIV-1 activity.
Materials and methods: PSC-RANTES nanoparticles formulated via a double emulsion process and characterized in both in vitro and ex vivo systems to determine PSC-RANTES release rate, nanoparticle tissue permeation, and anti-HIV bioactivity.
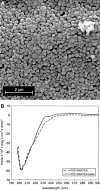
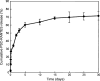
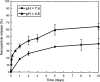
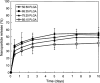
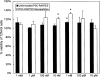
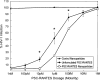
Results: Spherical, monodisperse (PDI = 0.098 +/- 0.054) PSC-RANTES nanoparticles (d = 256.58 +/- 19.57 nm) with an encapsulation efficiency of 82.23 +/- 8.35% were manufactured. In vitro release studies demonstrated a controlled release profile of PSC-RANTES (71.48 +/- 5.25% release). PSC-RANTES nanoparticle maintained comparable anti-HIV activity with unformulated PSC-RANTES in a HeLa cell-based system with an IC(50) of approximately 1pM. In an ex vivo cervical tissue model, PSC-RANTES nanoparticles displayed a fivefold increase in tissue uptake, enhanced tissue permeation, and significant localization at the basal layers of the epithelium over unformulated PSC-RANTES.
Conclusions: These results indicate that PSC-RANTES can readily be encapsulated into a PLGA nanoparticle drug delivery system, retain its anti-HIV-1 activity, and deliver PSC-RANTES to the target tissue. This is crucial for the success of this drug candidate as a topical microbicide product.
Figures

Similar articles
-
PSC-RANTES blocks R5 human immunodeficiency virus infection of Langerhans cells isolated from individuals with a variety of CCR5 diplotypes.J Virol. 2004 Jul;78(14):7602-9. doi: 10.1128/JVI.78.14.7602-7609.2004. J Virol. 2004. PMID: 15220435 Free PMC article.
-
Semisynthetic analogues of PSC-RANTES, a potent anti-HIV protein.Bioconjug Chem. 2008 Feb;19(2):480-9. doi: 10.1021/bc7003044. Epub 2008 Jan 8. Bioconjug Chem. 2008. PMID: 18179159
-
Noncovalent PEG Coating of Nanoparticle Drug Carriers Improves the Local Pharmacokinetics of Rectal Anti-HIV Microbicides.ACS Appl Mater Interfaces. 2018 Oct 17;10(41):34942-34953. doi: 10.1021/acsami.8b12214. Epub 2018 Oct 4. ACS Appl Mater Interfaces. 2018. PMID: 30234288
-
Biodegradable Particulate Carrier Formulation and Tuning for Targeted Drug Delivery.J Biomed Nanotechnol. 2015 Apr;11(4):555-77. doi: 10.1166/jbn.2015.2017. J Biomed Nanotechnol. 2015. PMID: 26310064 Review.
-
Nanoparticle systems as tools to improve drug delivery and therapeutic efficacy.J Biomed Mater Res A. 2013 Dec;101(12):3646-60. doi: 10.1002/jbm.a.34642. Epub 2013 Jul 22. J Biomed Mater Res A. 2013. PMID: 23878102 Review.
Cited by
-
Progress in antiretroviral drug delivery using nanotechnology.Int J Nanomedicine. 2010 Aug 9;5:533-47. Int J Nanomedicine. 2010. PMID: 20957115 Free PMC article. Review.
-
Recent Advances in the Excipients Used in Modified Release Vaginal Formulations.Materials (Basel). 2022 Jan 3;15(1):327. doi: 10.3390/ma15010327. Materials (Basel). 2022. PMID: 35009472 Free PMC article. Review.
-
Topical microbicides to prevent the transmission of HIV: formulation gaps and challenges.Drug Deliv Transl Res. 2011 Jun;1(3):194-200. doi: 10.1007/s13346-011-0034-2. Epub 2011 May 21. Drug Deliv Transl Res. 2011. PMID: 21766050 Free PMC article.
-
A review of nanotechnological approaches for the prophylaxis of HIV/AIDS.Biomaterials. 2013 Aug;34(26):6202-28. doi: 10.1016/j.biomaterials.2013.05.012. Epub 2013 May 28. Biomaterials. 2013. PMID: 23726227 Free PMC article. Review.
-
Drug synergy of tenofovir and nanoparticle-based antiretrovirals for HIV prophylaxis.PLoS One. 2013 Apr 22;8(4):e61416. doi: 10.1371/journal.pone.0061416. Print 2013. PLoS One. 2013. PMID: 23630586 Free PMC article.
References
-
- UNAIDS. AIDS Epidemic Update. 2007
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

