Related effects of cell adaptation to serum-free conditions on murine EPO production and glycosylation by CHO cells
- PMID: 19002864
- PMCID: PMC3449423
- DOI: 10.1007/s10616-006-9039-y
Related effects of cell adaptation to serum-free conditions on murine EPO production and glycosylation by CHO cells
Abstract
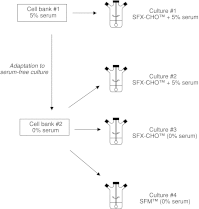
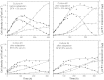
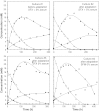
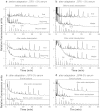
The necessity to perform serum-free cultures to produce recombinant glycoproteins generally requires an adaptation procedure of the cell line to new environmental conditions, which may therefore induce quantitative and qualitative effects on the product, particularly on its glycosylation. In previous studies, desialylation of EPO produced by CHO cells was shown to be dependent on the presence of serum in the medium. In this paper, to discriminate between the effects of the adaptation procedure to serum-free medium and the effects of the absence of serum on EPO production and glycosylation, adapted and non-adapted CHO cells were grown in serum-free and serum-containing media. The main kinetics of CHO cells were determined over batch processes as well as the glycosylation patterns of produced EPO by HPCE-LIF. A reversible decrease in EPO production was observed when cells were adapted to SFX-CHO(TM) medium, as the same cells partially recovered their production capacity when cultivated in serum-containing medium or in the enriched SFM(TM) serum-free medium. More interestingly, EPO desialylation that was not observed in both serum-free media was restored if the serum-independent cells were recultured in presence of serum. In the same way, while the serum-independent cells did not release a sialidase activity in both serum-free media, a significant activity was recovered when serum was added. In fact, the cell adaptation process to serum-free conditions did not specifically affect the sialidase release and the cellular mechanism of protein desialylation, which appeared to be mainly related to the presence of serum for both adapted and non-adapted cells.
Figures
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

