Retrograde adenoviral vector targeting of nociresponsive pontospinal noradrenergic neurons in the rat in vivo
- PMID: 19003793
- PMCID: PMC2659361
- DOI: 10.1002/cne.21879
Retrograde adenoviral vector targeting of nociresponsive pontospinal noradrenergic neurons in the rat in vivo
Abstract
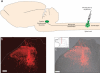
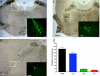
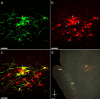
The spinal dorsal horn receives a dense innervation of noradrenaline-containing fibers that originate from pontine neurons in the A5, locus coeruleus (LC), and A7 cell groups. These pontospinal neurons are believed to constitute a component of the endogenous analgesic system. We used an adenoviral vector with a catecholaminergic-selective promoter (AVV-PRS) to retrogradely label the noradrenergic neurons projecting to the lumbar (L4-L5) dorsal horn with enhanced green fluorescent protein (EGFP) or monomeric red fluorescent protein (mRFP). Retrogradely labeled neurons (145 +/- 12, n = 14) were found in A5-12%, LC-80% and A7-8% after injection of AVV-PRS-EGFP to the dorsal horn of L4-L5. These neurons were immunopositive for dopamine beta-hydroxylase, indicating that they were catecholaminergic. Retrograde labeling was optimal 7 days after injection, persisted for over 4 weeks, and was dependent on viral vector titer. The spinal topography of the noradrenergic projection was examined using EGFP- and mRFP-expressing adenoviral vectors. Pontospinal neurons provide bilateral innervation of the cord and there was little overlap in the distribution of neurons projecting to the cervical and lumbar regions. The axonal arbor of the pontospinal neurons was visualized with GFP immunocytochemistry to show projections to the inferior olive, cerebellum, thalamus, and cortex but not to the hippocampus or caudate putamen. Formalin testing evoked c-fos expression in these pontospinal neurons, suggesting that they were nociresponsive (A5-21%, LC-16%, and A7-26%, n = 8). Thus, we have developed a viral vector-based strategy to selectively, retrogradely target the pontospinal noradrenergic neurons that are likely to be involved in the descending control of nociception.
Figures
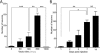




Similar articles
-
Retrograde viral vector-mediated inhibition of pontospinal noradrenergic neurons causes hyperalgesia in rats.J Neurosci. 2009 Oct 14;29(41):12855-64. doi: 10.1523/JNEUROSCI.1699-09.2009. J Neurosci. 2009. PMID: 19828800 Free PMC article.
-
Retrograde optogenetic characterization of the pontospinal module of the locus coeruleus with a canine adenoviral vector.Brain Res. 2016 Jun 15;1641(Pt B):274-90. doi: 10.1016/j.brainres.2016.02.023. Epub 2016 Feb 19. Brain Res. 2016. PMID: 26903420 Free PMC article.
-
Demonstration of two separate descending noradrenergic pathways to the rat spinal cord: evidence for an intragriseal trajectory of locus coeruleus axons in the superficial layers of the dorsal horn.J Comp Neurol. 1990 Jan 22;291(4):553-82. doi: 10.1002/cne.902910406. J Comp Neurol. 1990. PMID: 2329191
-
The projections of locus coeruleus neurons to the spinal cord.Prog Brain Res. 1991;88:123-41. doi: 10.1016/s0079-6123(08)63803-0. Prog Brain Res. 1991. PMID: 1813919 Review.
-
The noradrenergic pain regulation system: a potential target for pain therapy.Eur J Pharmacol. 2013 Sep 15;716(1-3):2-7. doi: 10.1016/j.ejphar.2013.01.067. Epub 2013 Mar 13. Eur J Pharmacol. 2013. PMID: 23500194 Review.
Cited by
-
Spinal astrocytes in superficial laminae gate brainstem descending control of mechanosensory hypersensitivity.Nat Neurosci. 2020 Nov;23(11):1376-1387. doi: 10.1038/s41593-020-00713-4. Epub 2020 Oct 5. Nat Neurosci. 2020. PMID: 33020652
-
The role of medial prefrontal cortex projections to locus ceruleus in mediating the sex differences in behavior in mice with inflammatory pain.FASEB J. 2021 Jul;35(7):e21747. doi: 10.1096/fj.202100319RR. FASEB J. 2021. PMID: 34151467 Free PMC article.
-
Functional dichotomy in spinal- vs prefrontal-projecting locus coeruleus modules splits descending noradrenergic analgesia from ascending aversion and anxiety in rats.Elife. 2017 Oct 13;6:e29808. doi: 10.7554/eLife.29808. Elife. 2017. PMID: 29027903 Free PMC article.
-
Retrograde viral vector-mediated inhibition of pontospinal noradrenergic neurons causes hyperalgesia in rats.J Neurosci. 2009 Oct 14;29(41):12855-64. doi: 10.1523/JNEUROSCI.1699-09.2009. J Neurosci. 2009. PMID: 19828800 Free PMC article.
-
Endogenous analgesic action of the pontospinal noradrenergic system spatially restricts and temporally delays the progression of neuropathic pain following tibial nerve injury.Pain. 2013 Sep;154(9):1680-1690. doi: 10.1016/j.pain.2013.05.010. Epub 2013 May 14. Pain. 2013. PMID: 23707289 Free PMC article.
References
-
- Abercrombie M. Estimation of nuclear population from microtome sections. Anat Rec. 1946;94:239–247. - PubMed
-
- Akli S, Caillaud C, Vigne E, Stratford-Perricaudet LD, Poenaru L, Perricaudet M, Kahn A, Peschanski MR. Transfer of a foreign gene into the brain using adenovirus vectors. Nat Genet. 1993;3:224–228. - PubMed
-
- Baulmann J, Spitznagel H, Herdegen T, Unger T, Culman J. Tachykinin receptor inhibition and c-Fos expression in the rat brain following formalin-induced pain. Neuroscience. 2000;95:813–820. - PubMed
-
- Bewig B, Schmidt WE. Accelerated titering of adenoviruses. Biotechniques. 2000;28:870–873. - PubMed
-
- Brunet JF, Pattyn A. Phox2 genes — from patterning to connectivity. Curr Opin Genet Dev. 2002;12:435–440. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

