MAGE-B2 autoantibody: a new biomarker for pediatric systemic lupus erythematosus
- PMID: 19004030
- PMCID: PMC2765927
- DOI: 10.3899/jrheum.080333
MAGE-B2 autoantibody: a new biomarker for pediatric systemic lupus erythematosus
Abstract
Objective: Melanoma-associated antigen gene B2 (MAGE-B2) encodes an embryonic antigen normally silenced after birth except in testis and placenta. We identified the MAGE-B2 gene and autoantibodies in pediatric patients with systemic lupus erythematosus (SLE) glomerulonephritis. We investigated the prevalence of MAGE-B2 autoantibodies in association with active SLE, to determine a pathogenetic role of MAGE-B2 protein through its distribution in cells and tissues.
Methods: A cross-sectional study analyzed the frequency of MAGE-B2 autoantibodies in 40 patients with pediatric SLE, 23 adult controls, and 16 patients with pediatric juvenile rheumatoid arthritis (JRA) using Western blots containing recombinant MAGE-B2. SLE Disease Activity Index 2000 (SLEDAI-2K) and British Isles Lupus Assessment Group (BILAG) index measured SLE disease activity. Tissue distribution of MAGE-B2 protein was assessed by immunohistochemistry, immunofluorescence, and Western blots.
Results: Seventeen (43%) of 40 pediatric SLE patients had MAGE-B2 autoantibodies as compared to 0 of 16 JRA patients and 2 of 23 adult controls. SLE disease activity was significantly higher in MAGE-B2 autoantibody-positive versus autoantibody-negative patients (SLEDAI-2K, mean 10.9 vs 5.2, p = 0.013; BILAG, mean 15.3 vs 6.3, p = 0.023). Active nephritis was more prevalent (60% vs 24%) in MAGE-B2 autoantibody-positive than autoantibody-negative SLE patients. MAGE-B2 protein was visualized in SLE kidney proximal convoluted tubules and in tumor epithelial cells, but not in lymphoblastoid cells.
Conclusion: MAGE-B2 autoantibody appears to be a clinically relevant biomarker for pediatric SLE disease activity and nephritis.
Figures
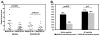




Similar articles
-
Comprehensive Longitudinal Surveillance of the IgG Autoantibody Repertoire in Established Systemic Lupus Erythematosus.Arthritis Rheumatol. 2019 May;71(5):736-743. doi: 10.1002/art.40788. Epub 2019 Mar 24. Arthritis Rheumatol. 2019. PMID: 30474930
-
A study on anti-mannose binding lectin (anti-MBL) antibodies and serum MBL levels in Indian systemic lupus erythematosus patients.Rheumatol Int. 2013 May;33(5):1193-9. doi: 10.1007/s00296-012-2519-9. Epub 2012 Sep 13. Rheumatol Int. 2013. PMID: 22971892
-
Comparison between multiplex assays for autoantibody detection in systemic lupus erythematosus.J Immunol Methods. 2010 Jun 30;358(1-2):75-80. doi: 10.1016/j.jim.2010.04.005. Epub 2010 May 9. J Immunol Methods. 2010. PMID: 20438730
-
Neutrophil Extracellular Trap Mitochondrial DNA and Its Autoantibody in Systemic Lupus Erythematosus and a Proof-of-Concept Trial of Metformin.Arthritis Rheumatol. 2015 Dec;67(12):3190-200. doi: 10.1002/art.39296. Arthritis Rheumatol. 2015. PMID: 26245802 Clinical Trial.
-
Anti-GAPDH Autoantibody Is Associated with Increased Disease Activity and Intracranial Pressure in Systemic Lupus Erythematosus.J Immunol Res. 2019 Mar 31;2019:7430780. doi: 10.1155/2019/7430780. eCollection 2019. J Immunol Res. 2019. PMID: 31049359 Free PMC article.
Cited by
-
Biomarkers for lupus nephritis: a critical appraisal.J Biomed Biotechnol. 2010;2010:638413. doi: 10.1155/2010/638413. Epub 2010 Apr 19. J Biomed Biotechnol. 2010. PMID: 20414362 Free PMC article. Review.
-
A Comprehensive Guide to the MAGE Family of Ubiquitin Ligases.J Mol Biol. 2017 Apr 21;429(8):1114-1142. doi: 10.1016/j.jmb.2017.03.005. Epub 2017 Mar 11. J Mol Biol. 2017. PMID: 28300603 Free PMC article. Review.
-
Serum tests for colorectal cancer screening.Mol Diagn Ther. 2011 Jun 1;15(3):129-41. doi: 10.1007/BF03256403. Mol Diagn Ther. 2011. PMID: 21766904
-
Emerging roles of the MAGE protein family in stress response pathways.J Biol Chem. 2020 Nov 20;295(47):16121-16155. doi: 10.1074/jbc.REV120.008029. Epub 2020 Sep 13. J Biol Chem. 2020. PMID: 32921631 Free PMC article. Review.
-
Role(s) of G3BPs in Human Pathogenesis.J Pharmacol Exp Ther. 2023 Oct;387(1):100-110. doi: 10.1124/jpet.122.001538. Epub 2023 Jul 19. J Pharmacol Exp Ther. 2023. PMID: 37468286 Free PMC article. Review.
References
-
- Petty RE, Laxer RM. Systemic Lupus Erythematosus. In: Cassidy J, Petty R, Laxer R, Lindsley C, editors. Textbook of Pediatric Rheumatology. 5. Philadelphia: Elselvier, Inc; 2005. pp. 342–383.
-
- Adorini L, Sinigaglia F. Pathogenesis and immunotherapy of autoimmune diseases. Trends Immunol. 1997;18:209–11. - PubMed
-
- McCurdy DK, Tai L-Q, Nguyen J, Wang Z, Yang H-M, Udar N, et al. MAGE Xp-2: A member of the MAGE gene family isolated from an expression library using systemic lupus erythematosus sera. Mol Genet Metabol. 1998;63:3–13. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
