Correlation of fragile histidine triad (Fhit) protein structural features with effector interactions and biological functions
- PMID: 19004824
- PMCID: PMC2613607
- DOI: 10.1074/jbc.M806638200
Correlation of fragile histidine triad (Fhit) protein structural features with effector interactions and biological functions
Abstract
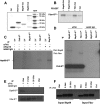
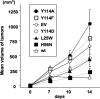
We have previously shown that Fhit tumor suppressor protein interacts with Hsp60 chaperone machinery and ferredoxin reductase (Fdxr) protein. Fhit-effector interactions are associated with a Fhit-dependent increase in Fdxr stability, followed by generation of reactive oxygen species and apoptosis induction under conditions of oxidative stress. To define Fhit structural features that affect interactions, downstream signaling, and biological outcomes, we used cancer cells expressing Fhit mutants with amino acid substitutions that alter enzymatic activity, enzyme substrate binding, or phosphorylation at tyrosine 114. Gastric cancer cell clones stably expressing mutants that do not bind substrate or cannot be phosphorylated showed decreased binding to Hsp60 and Fdxr and reduced mitochondrial localization. Expression of Fhit or mutants that bind interactor proteins results in oxidative damage and accumulation of cells in G(2)/M or sub-G(1) fractions after peroxide treatment; noninteracting mutants are defective in these biological effects. Gastric cancer clones expressing noncomplexing Fhit mutants show reduction of Fhit tumor suppressor activity, confirming that substrate binding, interaction with heat shock proteins, mitochondrial localization, and interaction with Fdxr are important for Fhit tumor suppressor function.
Figures

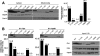



Similar articles
-
Characterization of the role of Fhit in suppression of DNA damage.Adv Biol Regul. 2013 Jan;53(1):77-85. doi: 10.1016/j.jbior.2012.10.003. Epub 2012 Oct 11. Adv Biol Regul. 2013. PMID: 23102829 Free PMC article.
-
Fhit-Fdxr interaction in the mitochondria: modulation of reactive oxygen species generation and apoptosis in cancer cells.Cell Death Dis. 2019 Feb 15;10(3):147. doi: 10.1038/s41419-019-1414-7. Cell Death Dis. 2019. PMID: 30770797 Free PMC article.
-
Fragile gene product, Fhit, in oxidative and replicative stress responses.Cancer Sci. 2009 Jul;100(7):1145-50. doi: 10.1111/j.1349-7006.2009.01168.x. Epub 2009 Mar 23. Cancer Sci. 2009. PMID: 19486340 Free PMC article. Review.
-
Activation state-dependent interaction between Gαq subunits and the Fhit tumor suppressor.Cell Commun Signal. 2013 Aug 15;11:59. doi: 10.1186/1478-811X-11-59. Cell Commun Signal. 2013. PMID: 23947369 Free PMC article.
-
Fhit tumor suppressor: guardian of the preneoplastic genome.Future Oncol. 2008 Dec;4(6):815-24. doi: 10.2217/14796694.4.6.815. Future Oncol. 2008. PMID: 19086848 Free PMC article. Review.
Cited by
-
FHIT and C-MYC expression in cervical histology and cytology as biomarkers for detecting high-grade intraepithelial neoplasia in human papillomavirus-positive women.Cancer Biomark. 2020;27(3):303-312. doi: 10.3233/CBM-182232. Cancer Biomark. 2020. PMID: 31771040 Free PMC article.
-
Identification of Fhit as a post-transcriptional effector of Thymidine Kinase 1 expression.Biochim Biophys Acta Gene Regul Mech. 2017 Mar;1860(3):374-382. doi: 10.1016/j.bbagrm.2017.01.005. Epub 2017 Jan 14. Biochim Biophys Acta Gene Regul Mech. 2017. PMID: 28093273 Free PMC article.
-
Initiation of genome instability and preneoplastic processes through loss of Fhit expression.PLoS Genet. 2012;8(11):e1003077. doi: 10.1371/journal.pgen.1003077. Epub 2012 Nov 29. PLoS Genet. 2012. PMID: 23209436 Free PMC article.
-
Abnormal FHIT protein expression may be correlated with poor prognosis in gastric cancer: a meta-analysis.Tumour Biol. 2014 Jul;35(7):6815-21. doi: 10.1007/s13277-014-1936-7. Epub 2014 Apr 12. Tumour Biol. 2014. PMID: 24729090
-
A knockdown with smoke model reveals FHIT as a repressor of Heme oxygenase 1.Cell Cycle. 2014;13(18):2913-30. doi: 10.4161/15384101.2014.946858. Cell Cycle. 2014. PMID: 25486479 Free PMC article.
References
-
- Huebner, K., and Croce, C. M. (2001) Nat. Rev. Cancer 1 214-221 - PubMed
-
- Barnes, L. D., Garrison, P. N., Siprashvili, Z., Guranowski, A., Robinson, A. K., Ingram, S. W., Croce, C. M., Ohta, M., and Huebner, K. (1996) Biochemistry 35 11529-11535 - PubMed
-
- Ji, L., Fang, B., Yen, N., Fong, K., Minna, J. D., and Roth, J. A. (1999) Cancer Res. 59 3333-3339 - PubMed
-
- Ishii, H., Dumon, K. R., Vecchione, A., Trapasso, F., Mimori, K., Alder, H., Mori, M., Sozzi, G., Baffa, R., Huebner, K., and Croce, C. M. (2001) Cancer Res. 61 1578-1584 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

