Impaired mitochondria-dependent vasodilation in cerebral arteries of Zucker obese rats with insulin resistance
- PMID: 19005015
- PMCID: PMC2643987
- DOI: 10.1152/ajpregu.90656.2008
Impaired mitochondria-dependent vasodilation in cerebral arteries of Zucker obese rats with insulin resistance
Abstract
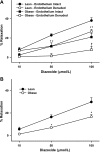
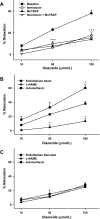
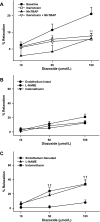
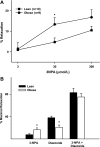
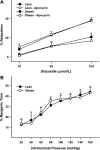
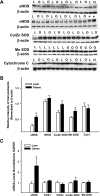
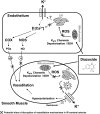
Mitochondria affect cerebrovascular tone by activation of mitochondrial ATP-sensitive K+ (K ATP) channels and generation of reactive oxygen species (ROS). Insulin resistance accompanying obesity causes mitochondrial dysfunction, but the consequences on the cerebral circulation have not been fully identified. We evaluated the mitochondrial effects of diazoxide, a putative mitochondrial K ATP channel activator, on cerebral arteries of Zucker obese (ZO) rats with insulin resistance and lean (ZL) controls. Diameter measurements showed diminished diazoxide-induced vasodilation in ZO compared with ZL rats. Maximal relaxation was 38 +/- 3% in ZL vs. 21 +/- 4% in ZO rats (P < 0.05). Iberiotoxin, a Ca2+-activated K+ channel inhibitor, or manganese(III) tetrakis(4-benzoic acid)porphyrin chloride, an SOD mimetic, or endothelial denudation diminished vasodilation to diazoxide, implicating Ca2+-activated K+ channels, ROS, and endothelial factors in vasodilation. Inhibition of nitric oxide synthase (NOS) in ZL rats diminished diazoxide-induced vasodilation in intact arteries, but vasodilation was unaffected in endothelium-denuded arteries. In contrast, NOS inhibition in ZO rats enhanced vasodilation in endothelium-denuded arteries, but intact arteries were unaffected, suggesting that activity of endothelial NOS was abolished, whereas factors derived from nonendothelial NOS promoted vasoconstriction. Fluorescence microscopy showed decreased mitochondrial depolarization, ROS production, and nitric oxide generation in response to diazoxide in ZO arteries. Protein and mRNA measurements revealed increased expression of endothelial NOS and SODs in ZO arteries. Thus, cerebrovascular dilation to mitochondria-derived factors involves integration of endothelial and smooth muscle mechanisms. Furthermore, mitochondria-mediated vasodilation was diminished in ZO rats due to impaired mitochondrial K(ATP) channel activation, diminished mitochondrial ROS generation, increased ROS scavenging, and abnormal NOS activity.
Figures

Similar articles
-
Depolarization of mitochondria in endothelial cells promotes cerebral artery vasodilation by activation of nitric oxide synthase.Arterioscler Thromb Vasc Biol. 2013 Apr;33(4):752-9. doi: 10.1161/ATVBAHA.112.300560. Epub 2013 Jan 17. Arterioscler Thromb Vasc Biol. 2013. PMID: 23329133 Free PMC article.
-
Diversity of mitochondria-dependent dilator mechanisms in vascular smooth muscle of cerebral arteries from normal and insulin-resistant rats.Am J Physiol Heart Circ Physiol. 2014 Aug 15;307(4):H493-503. doi: 10.1152/ajpheart.00091.2014. Am J Physiol Heart Circ Physiol. 2014. PMID: 24929852 Free PMC article.
-
Impaired insulin-induced vasodilation in small coronary arteries of Zucker obese rats is mediated by reactive oxygen species.Am J Physiol Heart Circ Physiol. 2005 Feb;288(2):H854-60. doi: 10.1152/ajpheart.00715.2004. Am J Physiol Heart Circ Physiol. 2005. PMID: 15650157
-
Calcium-activated potassium channels and endothelial dysfunction: therapeutic options?Br J Pharmacol. 2009 Feb;156(4):545-62. doi: 10.1111/j.1476-5381.2009.00052.x. Epub 2009 Jan 29. Br J Pharmacol. 2009. PMID: 19187341 Free PMC article. Review.
-
The effects of obesity on the cerebral vasculature.Curr Vasc Pharmacol. 2014 May;12(3):462-72. doi: 10.2174/1570161112666140423222411. Curr Vasc Pharmacol. 2014. PMID: 24846235 Free PMC article. Review.
Cited by
-
Cyclooxygenase-derived vasoconstriction restrains hypoxia-mediated cerebral vasodilation in young adults with metabolic syndrome.Am J Physiol Heart Circ Physiol. 2014 Jan 15;306(2):H261-9. doi: 10.1152/ajpheart.00709.2013. Epub 2013 Nov 8. Am J Physiol Heart Circ Physiol. 2014. PMID: 24213610 Free PMC article. Clinical Trial.
-
Impaired Mitochondrial Respiration in Large Cerebral Arteries of Rats with Type 2 Diabetes.J Vasc Res. 2017;54(1):1-12. doi: 10.1159/000454812. Epub 2017 Jan 18. J Vasc Res. 2017. PMID: 28095372 Free PMC article.
-
Insulin-resistant brain state: the culprit in sporadic Alzheimer's disease?Ageing Res Rev. 2011 Apr;10(2):264-73. doi: 10.1016/j.arr.2011.01.001. Epub 2011 Jan 22. Ageing Res Rev. 2011. PMID: 21262392 Free PMC article. Review.
-
Impaired response to exercise intervention in the vasculature in metabolic syndrome.Diab Vasc Dis Res. 2013 May;10(3):222-38. doi: 10.1177/1479164112459664. Epub 2012 Nov 16. Diab Vasc Dis Res. 2013. PMID: 23162060 Free PMC article.
-
Neuroprotection from protein misfolding in cerebral hypoperfusion concurrent with metabolic syndrome. A translational perspective.Front Neurosci. 2023 Aug 15;17:1215041. doi: 10.3389/fnins.2023.1215041. eCollection 2023. Front Neurosci. 2023. PMID: 37650104 Free PMC article. Review.
References
-
- Arenillas JF, Moro MA, Davalos A. The metabolic syndrome and stroke: potential treatment approaches. Stroke 38: 2196–2203, 2007. - PubMed
-
- Bray GA The Zucker-fatty rat: a review. Fed Proc 36: 148–153, 1977. - PubMed
-
- Busija DW, Katakam P, Rajapakse NC, Kis B, Grover G, Domoki F, Bari F. Effects of ATP-sensitive potassium channel activators diazoxide and BMS-191095 on membrane potential and reactive oxygen species production in isolated piglet mitochondria. Brain Res Bull 66: 85–90, 2005. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

