Deciliation is associated with dramatic remodeling of epithelial cell junctions and surface domains
- PMID: 19005211
- PMCID: PMC2613083
- DOI: 10.1091/mbc.e08-07-0741
Deciliation is associated with dramatic remodeling of epithelial cell junctions and surface domains
Abstract
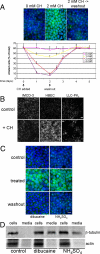
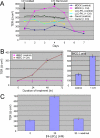
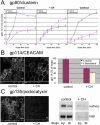
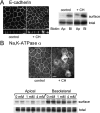
Stress-induced shedding of motile cilia (autotomy) has been documented in diverse organisms and likely represents a conserved cellular reaction. However, little is known about whether primary cilia are shed from mammalian epithelial cells and what impact deciliation has on polarized cellular organization. We show that several chemically distinct agents trigger autotomy in epithelial cells. Surprisingly, deciliation is associated with a significant, but reversible increase in transepithelial resistance. This reflects substantial reductions in tight junction proteins associated with "leaky" nephron segments (e.g., claudin-2). At the same time, apical trafficking of gp80/clusterin and gp114/CEACAM becomes randomized, basal-lateral delivery of Na,K-ATPase is reduced, and expression of the nonciliary apical protein gp135/podocalyxin is greatly decreased. However, ciliogenesis-impaired MDCK cells do not undergo continual junction remodeling, and mature cilia are not required for autotomy-associated remodeling events. Deciliation and epithelial remodeling may be mechanistically linked processes, because RNAi-mediated reduction of Exocyst subunit Sec6 inhibits ciliary shedding and specifically blocks deciliation-associated down-regulation of claudin-2 and gp135. We propose that ciliary autotomy represents a signaling pathway that impacts the organization and function of polarized epithelial cells.
Figures



Similar articles
-
Rab27 effector Slp2-a transports the apical signaling molecule podocalyxin to the apical surface of MDCK II cells and regulates claudin-2 expression.Mol Biol Cell. 2012 Aug;23(16):3229-39. doi: 10.1091/mbc.E12-02-0104. Epub 2012 Jul 5. Mol Biol Cell. 2012. PMID: 22767581 Free PMC article.
-
Juxtacrine activation of EGFR regulates claudin expression and increases transepithelial resistance.Am J Physiol Cell Physiol. 2007 Nov;293(5):C1660-8. doi: 10.1152/ajpcell.00274.2007. Epub 2007 Sep 13. Am J Physiol Cell Physiol. 2007. PMID: 17855771
-
Galectin-8 regulates targeting of Gp135/podocalyxin and lumen formation at the apical surface of renal epithelial cells.FASEB J. 2017 Nov;31(11):4917-4927. doi: 10.1096/fj.201601386R. Epub 2017 Jul 26. FASEB J. 2017. PMID: 28747404
-
Cellular deflagellation.Int Rev Cytol. 2004;233:47-91. doi: 10.1016/S0074-7696(04)33002-0. Int Rev Cytol. 2004. PMID: 15037362 Review.
-
Tight junction: a co-ordinator of cell signalling and membrane trafficking.Biol Cell. 2005 Aug;97(8):659-65. doi: 10.1042/BC20040147. Biol Cell. 2005. PMID: 16033326 Review.
Cited by
-
Carcinogens induce loss of the primary cilium in human renal proximal tubular epithelial cells independently of effects on the cell cycle.Am J Physiol Renal Physiol. 2012 Apr 15;302(8):F905-16. doi: 10.1152/ajprenal.00427.2011. Epub 2012 Jan 18. Am J Physiol Renal Physiol. 2012. PMID: 22262483 Free PMC article.
-
A Rab8 guanine nucleotide exchange factor-effector interaction network regulates primary ciliogenesis.J Biol Chem. 2012 May 4;287(19):15602-9. doi: 10.1074/jbc.M111.333245. Epub 2012 Mar 19. J Biol Chem. 2012. PMID: 22433857 Free PMC article.
-
Brain heterotopia formation by ciliopathic breakdown of neuroepithelial and blood-cerebrospinal fluid barriers.Brain Pathol. 2023 Jul;33(4):e13148. doi: 10.1111/bpa.13148. Epub 2023 Jan 9. Brain Pathol. 2023. PMID: 36623505 Free PMC article.
-
Fluid shear stress-induced TGF-β/ALK5 signaling in renal epithelial cells is modulated by MEK1/2.Cell Mol Life Sci. 2017 Jun;74(12):2283-2298. doi: 10.1007/s00018-017-2460-x. Epub 2017 Feb 6. Cell Mol Life Sci. 2017. PMID: 28168444 Free PMC article.
-
Oligodendroglial primary cilium heterogeneity during development and demyelination/remyelination.Front Cell Neurosci. 2022 Nov 24;16:1049468. doi: 10.3389/fncel.2022.1049468. eCollection 2022. Front Cell Neurosci. 2022. PMID: 36505511 Free PMC article.
References
-
- Balda M. S., Gonzalez-Mariscal L., Contreras R. G., Macias-Silva M., Torres-Marquez M. E., Garcia-Sainz J. A., Cereijido M. Assembly and sealing of tight junctions: possible participation of G-proteins, phospholipase C, protein kinase C and calmodulin. J. Membr. Biol. 1991;122:193–202. - PubMed
-
- Bergesse J. R., Domenech C. E., Balegno H. F. Chloral hydrate inhibition in vitro of ATPase in membrane of rat erythrocytes and in microsomes of dog kidney external medulla. Biochem. Pharmacol. 1983;32:3221–3225. - PubMed
-
- Blum J. J. Existence of a breaking point in cilia and flagella. J. Theor. Biol. 1971;33:257–263. - PubMed
-
- Burdett I.D. Internalisation of desmosomes and their entry into the endocytic pathway via late endosomes in MDCK cells. Possible mechanisms for the modulation of cell adhesion by desmosomes during development. J. Cell Sci. 1993;106(Pt 4):1115–1130. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

