Ceruloplasmin alters the tissue disposition and neurotoxicity of manganese, but not its loading onto transferrin
- PMID: 19005224
- PMCID: PMC2735423
- DOI: 10.1093/toxsci/kfn231
Ceruloplasmin alters the tissue disposition and neurotoxicity of manganese, but not its loading onto transferrin
Abstract
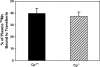
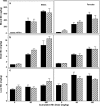
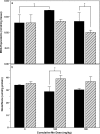
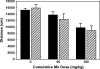
Manganese (Mn) is a redox-active element, and whereas its uptake, disposition, and toxicity in mammals may depend in part on its oxidation state, the proteins affecting manganese oxidation state and speciation in vivo are not well known. Studies have suggested that the oxidase protein ceruloplasmin (Cp) mediates iron and manganese oxidation and loading onto plasma transferrin (Tf), as well as cellular iron efflux. We hypothesized that ceruloplasmin may also affect the tissue distribution and eventual neurotoxicity of manganese. To test this, aceruloplasminemic versus wild-type mice were treated with a single i.p. (54)Mn tracer dose, or elevated levels of manganese subchronically (0, 7.5, or 15 mg Mn/kg s.c., three doses per week for 4 weeks), and evaluated for transferrin-bound manganese, blood manganese partitioning, tissue manganese disposition, and levels of brain glutathione, thiobarbituric acid reactive substances (TBARS), and protein carbonyls as measures of oxidative stress, and open arena activity. Results show that ceruloplasmin does not play a role in the loading of manganese onto plasma transferrin in vivo, or in the partitioning of manganese between the plasma and cellular fractions of whole blood. Ceruloplasmin did, however, affect the retention of manganese in blood and its distribution to tissues, most notably kidney and to a lesser extent brain and lung. Results also indicate that ceruloplasmin interacted with chronic elevated manganese exposures to produce greater levels of brain oxidative stress. These results provide evidence that metal oxidase proteins play an important role in altering neurotoxicity arising from elevated manganese exposures.
Figures

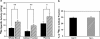
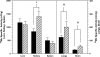
Similar articles
-
Increase in Fe3+/Fe2+ ratio and iron-induced oxidative stress in Eales disease and presence of ferrous iron in circulating transferrin.Curr Eye Res. 2007 Jul-Aug;32(7-8):677-83. doi: 10.1080/02713680701486402. Curr Eye Res. 2007. PMID: 17852192
-
Investigating the role of transferrin in the distribution of iron, manganese, copper, and zinc.J Biol Inorg Chem. 2014 Aug;19(6):869-77. doi: 10.1007/s00775-014-1118-5. Epub 2014 Feb 25. J Biol Inorg Chem. 2014. PMID: 24567067 Free PMC article.
-
Copper transport and metabolism are normal in aceruloplasminemic mice.J Biol Chem. 2001 Sep 28;276(39):36857-61. doi: 10.1074/jbc.M105361200. Epub 2001 Jul 18. J Biol Chem. 2001. PMID: 11461924
-
Pharmacokinetic data needs to support risk assessments for inhaled and ingested manganese.Neurotoxicology. 1999 Apr-Jun;20(2-3):161-71. Neurotoxicology. 1999. PMID: 10385880 Review.
-
Manganese Accumulation in the Brain via Various Transporters and Its Neurotoxicity Mechanisms.Molecules. 2020 Dec 12;25(24):5880. doi: 10.3390/molecules25245880. Molecules. 2020. PMID: 33322668 Free PMC article. Review.
Cited by
-
Manganese (Mn) and iron (Fe): interdependency of transport and regulation.Neurotox Res. 2010 Aug;18(2):124-31. doi: 10.1007/s12640-009-9130-1. Epub 2009 Nov 18. Neurotox Res. 2010. PMID: 19921534 Free PMC article. Review.
-
The biology of mammalian multi-copper ferroxidases.Biometals. 2023 Apr;36(2):263-281. doi: 10.1007/s10534-022-00370-z. Epub 2022 Feb 15. Biometals. 2023. PMID: 35167013 Free PMC article. Review.
-
Altered metal metabolism in patients with HCV-related cirrhosis and hepatic encephalopathy.Metab Brain Dis. 2015 Dec;30(6):1445-52. doi: 10.1007/s11011-015-9721-x. Epub 2015 Aug 27. Metab Brain Dis. 2015. PMID: 26307419
-
Redox dynamics of manganese as a mitochondrial life-death switch.Biochem Biophys Res Commun. 2017 Jan 15;482(3):388-398. doi: 10.1016/j.bbrc.2016.10.126. Epub 2017 Feb 3. Biochem Biophys Res Commun. 2017. PMID: 28212723 Free PMC article. Review.
-
Manganese flux across the blood-brain barrier.Neuromolecular Med. 2009;11(4):297-310. doi: 10.1007/s12017-009-8101-2. Epub 2009 Nov 10. Neuromolecular Med. 2009. PMID: 19902387 Review.
References
-
- Abe K, Chiba Y, Nishida Y. Facile uptake of manganese(III) by apotransferrin: Possible origin of manganism. Z. Naturforsch. 2008;63:154–156. - PubMed
-
- Aisen P, Aasa R, Redfield AG. The chromium, manganese, and cobalt complexes of transferrin. J. Biol. Chem. 1969;244:4628–4633. - PubMed
-
- Ali SF, Duhart HM, Newport GD, Lipe GW, Slikker W., Jr Manganese-induced reactive oxygen species: Comparison between Mn+2 and Mn+3. Neurodegeneration. 1995;4:329–334. - PubMed
-
- Ali SS, Xiong C, Lucero J, Behrens MM, Dugan LL, Quick KL. Gender differences in free radical homeostasis during aging: Shorter-lived female C57BL6 mice have increased oxidative stress. Aging Cell. 2006;5:565–574. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

