Tandem leader proteases of Grapevine leafroll-associated virus-2: host-specific functions in the infection cycle
- PMID: 19007962
- PMCID: PMC7103369
- DOI: 10.1016/j.virol.2008.09.035
Tandem leader proteases of Grapevine leafroll-associated virus-2: host-specific functions in the infection cycle
Abstract
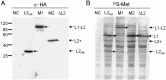
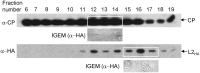
Several viruses in the genus Closterovirus including Grapevine leafroll-associated virus-2 (GLRaV-2), encode a tandem of papain-like leader proteases (L1 and L2) whose functional profiles remained largely uncharacterized. We generated a series of the full-length, reporter-tagged, clones of GLRaV-2 and demonstrated that they are systemically infectious upon agroinfection of an experimental host plant Nicotiana benthamiana. These clones and corresponding minireplicon derivatives were used to address L1 and L2 functions in GLRaV-2 infection cycle. It was found that the deletion of genome region encoding the entire L1-L2 tandem resulted in a ~100-fold reduction in minireplicon RNA accumulation. Five-fold reduction in RNA level was observed upon deletion of L1 coding region. In contrast, deletion of L2 coding region did not affect RNA accumulation. It was also found that the autocatalytic cleavage by L2 but not by L1 is essential for genome replication. Analysis of the corresponding mutants in the context of N. benthamiana infection launched by the full-length GLRaV-2 clone revealed that L1 or its coding region is essential for virus ability to establish infection, while L2 plays an accessory role in the viral systemic transport. Strikingly, when tagged minireplicon variants were used for the leaf agroinfiltration of the GLRaV-2 natural host, Vitis vinifera, deletion of either L1 or L2 resulted in a dramatic reduction of minireplicon ability to establish infection attesting to a host-specific requirement for tandem proteases in the virus infection cycle.
Figures


Similar articles
-
Biological, molecular, and serological studies of a novel strain of grapevine leafroll-associated virus 2.Virus Genes. 2011 Aug;43(1):102-10. doi: 10.1007/s11262-011-0607-7. Epub 2011 Apr 13. Virus Genes. 2011. PMID: 21487783
-
Development of a Minireplicon for Grapevine Leafroll-Associated Virus 1 and Genetic Analyses of Sequences in the 5' Non-Translated Region Required for Replication.Phytopathology. 2025 Aug;115(8):1065-1075. doi: 10.1094/PHYTO-12-24-0380-R. Epub 2025 Aug 6. Phytopathology. 2025. PMID: 40388575
-
Expression of the p24 silencing suppressor of Grapevine leafroll-associated virus 2 from Potato virus X or Barley stripe mosaic virus vector elicits hypersensitive responses in Nicotiana benthamiana.Plant Physiol Biochem. 2019 Sep;142:34-42. doi: 10.1016/j.plaphy.2019.06.033. Epub 2019 Jun 25. Plant Physiol Biochem. 2019. PMID: 31255907
-
Development of infectious cDNA clones of Grapevine leafroll-associated virus 3 and analyses of the 5' non-translated region for replication and virion formation.Virology. 2018 Oct;523:89-99. doi: 10.1016/j.virol.2018.07.023. Epub 2018 Aug 10. Virology. 2018. PMID: 30103103
-
Probing into the Effects of Grapevine Leafroll-Associated Viruses on the Physiology, Fruit Quality and Gene Expression of Grapes.Viruses. 2021 Mar 31;13(4):593. doi: 10.3390/v13040593. Viruses. 2021. PMID: 33807294 Free PMC article. Review.
Cited by
-
A plant virus evolved by acquiring multiple nonconserved genes to extend its host range.Proc Natl Acad Sci U S A. 2011 Oct 18;108(42):17366-71. doi: 10.1073/pnas.1113227108. Epub 2011 Oct 10. Proc Natl Acad Sci U S A. 2011. PMID: 21987809 Free PMC article.
-
Molecular Characterization of Divergent Closterovirus Isolates Infecting Ribes Species.Viruses. 2018 Jul 12;10(7):369. doi: 10.3390/v10070369. Viruses. 2018. PMID: 30002359 Free PMC article.
-
Infection with strains of Citrus tristeza virus does not exclude superinfection by other strains of the virus.J Virol. 2010 Feb;84(3):1314-25. doi: 10.1128/JVI.02075-09. Epub 2009 Nov 18. J Virol. 2010. PMID: 19923189 Free PMC article.
-
Biological, molecular, and serological studies of a novel strain of grapevine leafroll-associated virus 2.Virus Genes. 2011 Aug;43(1):102-10. doi: 10.1007/s11262-011-0607-7. Epub 2011 Apr 13. Virus Genes. 2011. PMID: 21487783
-
Mutagenesis of the catalytic and cleavage site residues of the hypovirus papain-like proteases p29 and p48 reveals alternative processing and contributions to optimal viral RNA accumulation.J Virol. 2014 Oct;88(20):11946-54. doi: 10.1128/JVI.01489-14. Epub 2014 Aug 6. J Virol. 2014. PMID: 25100848 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

