HDL remodeling during the acute phase response
- PMID: 19008529
- PMCID: PMC2760005
- DOI: 10.1161/ATVBAHA.108.178681
HDL remodeling during the acute phase response
Abstract
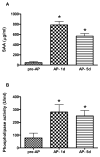
Objective: The purpose of this study was to examine the interactive action of serum amyloid A (SAA), group IIA secretory phospholipase A(2) (sPLA(2)-IIA), and cholesteryl ester transfer protein (CETP) on HDL remodeling and cholesterol efflux during the acute phase (AP) response elicited in humans after cardiac surgery.
Methods and results: Plasma was collected from patients before (pre-AP), 24 hours after (AP-1 d), and 5 days after cardiac surgery (AP-5 d). SAA levels were increased 16-fold in AP-1 d samples. The activity of sPLA(2)-IIA was increased from 77.7+/-38.3 U/mL (pre-AP) to 281.4+/-57.1 U/mL (AP-1 d; P<0.001). CETP mass and activity reduction was commensurate to the reduction of HDL cholesterol levels. The combined action of SAA, sPLA(2)-IIA, and CETP in vitro markedly remodeled HDL with the generation of lipid-poor apoA-I from both pre-AP and AP-1 d HDL. The net result of this remodeling was a relative preservation of ABCA1- and ABCG1-dependent cholesterol efflux during the acute phase response.
Conclusions: Our results show that the many and complex changes in plasma proteins during the acute phase response markedly remodel HDL with functional implications, particularly the relative retention of cholesterol efflux capacity.
Figures



Similar articles
-
ATP binding cassette G1-dependent cholesterol efflux during inflammation.J Lipid Res. 2011 Feb;52(2):345-53. doi: 10.1194/jlr.M012328. Epub 2010 Dec 7. J Lipid Res. 2011. PMID: 21138980 Free PMC article.
-
Serum amyloid A promotes cholesterol efflux mediated by scavenger receptor B-I.J Biol Chem. 2005 Oct 28;280(43):35890-5. doi: 10.1074/jbc.M505685200. Epub 2005 Aug 23. J Biol Chem. 2005. PMID: 16120612
-
HDL from CETP-deficient subjects shows enhanced ability to promote cholesterol efflux from macrophages in an apoE- and ABCG1-dependent pathway.J Clin Invest. 2006 May;116(5):1435-42. doi: 10.1172/JCI27602. J Clin Invest. 2006. PMID: 16670775 Free PMC article.
-
HDL cholesterol transport during inflammation.Curr Opin Lipidol. 2007 Apr;18(2):147-51. doi: 10.1097/MOL.0b013e328051b4fe. Curr Opin Lipidol. 2007. PMID: 17353662 Review.
-
High-density lipoprotein and the acute phase response.Curr Opin Endocrinol Diabetes Obes. 2010 Apr;17(2):156-60. doi: 10.1097/MED.0b013e328337278b. Curr Opin Endocrinol Diabetes Obes. 2010. PMID: 20177380 Free PMC article. Review.
Cited by
-
SAA does not induce cytokine production in physiological conditions.Cytokine. 2013 Feb;61(2):506-12. doi: 10.1016/j.cyto.2012.10.019. Epub 2012 Nov 17. Cytokine. 2013. PMID: 23165195 Free PMC article.
-
Role of Serum Amyloid A in Abdominal Aortic Aneurysm and Related Cardiovascular Diseases.Biomolecules. 2021 Dec 15;11(12):1883. doi: 10.3390/biom11121883. Biomolecules. 2021. PMID: 34944527 Free PMC article. Review.
-
Serum amyloid A binds to fibrin(ogen), promoting fibrin amyloid formation.Sci Rep. 2019 Feb 28;9(1):3102. doi: 10.1038/s41598-019-39056-x. Sci Rep. 2019. PMID: 30816210 Free PMC article.
-
An agomir of miR-144-3p accelerates plaque formation through impairing reverse cholesterol transport and promoting pro-inflammatory cytokine production.PLoS One. 2014 Apr 14;9(4):e94997. doi: 10.1371/journal.pone.0094997. eCollection 2014. PLoS One. 2014. PMID: 24733347 Free PMC article.
-
LpA-II:B:C:D:E: a new immunochemically-defined acute phase lipoprotein in humans.Lipids Health Dis. 2018 May 28;17(1):127. doi: 10.1186/s12944-018-0769-6. Lipids Health Dis. 2018. PMID: 29807532 Free PMC article.
References
-
- Cabana VG, Siegel JN, Sabesin SM. Effects of the acute phase response on the concentration and density distribution of plasma lipids and apolipoproteins. J Lipid Res. 1989;30:39–49. - PubMed
-
- Menschikowski M, Hagelgans A, Siegert G. Secretory phospholipase A2 of group IIA: is it an offensive or a defensive player during atherosclerosis and other inflammatory diseases? Prostaglandins Other Lipid Mediat. 2006;79:1–33. - PubMed
-
- Coetzee GA, Strachan AF, van der Westhuyzen DR, Hoppe HC, Jeenah MS, de Beer FC. Serum amyloid A-containing human high density lipoprotein 3. Density, size, and apolipoprotein composition. J Biol Chem. 1986;261:9644–9651. - PubMed
-
- de Beer FC, de Beer MC, van der Westhuyzen DR, Castellani LW, Lusis AJ, Swanson ME, Grass DS. Secretory non-pancreatic phospholipase A2: influence on lipoprotein metabolism. J Lipid Res. 1997;38:2232–2239. - PubMed
-
- Barter PJ. Hugh sinclair lecture: the regulation and remodelling of HDL by plasma factors. Atheroscler Suppl. 2002;3:39–47. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

