Aging mice exhibit a functional defect in mucosal dendritic cell response against an intracellular pathogen
- PMID: 19017989
- PMCID: PMC2676144
- DOI: 10.4049/jimmunol.181.11.7977
Aging mice exhibit a functional defect in mucosal dendritic cell response against an intracellular pathogen
Abstract
Down-regulation of the immune response in aging individuals puts this population at a potential risk against infectious agents. In-depth studies conducted in humans and mouse models have demonstrated that with increasing age, the T cell immune response against pathogens is compromised and response to vaccinations is subdued. In the present study, using a mouse model, we demonstrate that older animals exhibit greater susceptibility to Encephalitozoon cuniculi infection, and their ability to evoke an Ag-specific T cell response at the gut mucosal site is reduced. The dampening of T cell immunity was due to the defective priming by the dendritic cells (DC) isolated from the mucosal tissues of aging animals. When primed with DC from younger mice, T cells from older animals were able to exhibit an optimal Ag-specific response. The functional defect in DC from older mice can be attributed to a large extent to reduced IL-15 message in these cells, which can be reversed by addition of exogenous IL-15 to the cultures. IL-15 treatment led to optimal expression of costimulatory molecules (CD80 and CD86) on the surface of older DC and restored their ability to prime a T cell response against the pathogen. To our knowledge, this is the first report which demonstrates the inability of the DC population from aging animals to prime a robust T cell response against an infectious agent. Moreover, the observation that IL-15 treatment can reverse this defect has far-reaching implications in developing strategies to increase vaccination protocols for aging populations.
Figures
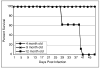
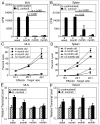
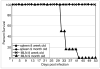

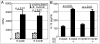


 9 month old DC + E. cuniculi,
9 month old DC + E. cuniculi,  9 month old DC + E. cuniculi + IL-15). Results are presented as mean ± SD of triplicates and experiment was performed twice with similar results.
9 month old DC + E. cuniculi + IL-15). Results are presented as mean ± SD of triplicates and experiment was performed twice with similar results.References
-
- Zheng B, Han S, Takahashi Y, Kelsoe G. Immunosenescence and germinal center reaction. Immunol Rev. 1997;160:63–77. - PubMed
-
- Song H, Price PW, Cerny J. Age-related changes in antibody repertoire: contribution from T cells. Immunol Rev. 1997;160:55–62. - PubMed
-
- Garcia GG, Miller RA. Differential tyrosine phosphorylation of zeta chain dimers in mouse CD4 T lymphocytes: effect of age. Cell Immunol. 1997;175:51–57. - PubMed
-
- Homann D, Teyton L, Oldstone MB. Differential regulation of antiviral T-cell immunity results in stable CD8+ but declining CD4+ T-cell memory. Nat Med. 2001;7:913–919. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

