A decade of Cdc14--a personal perspective. Delivered on 9 July 2007 at the 32nd FEBS Congress in Vienna, Austria
- PMID: 19021755
- PMCID: PMC2742780
- DOI: 10.1111/j.1742-4658.2008.06693.x
A decade of Cdc14--a personal perspective. Delivered on 9 July 2007 at the 32nd FEBS Congress in Vienna, Austria
Abstract
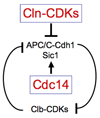
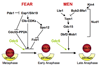
In budding yeast, the protein phosphatase Cdc14 is a key regulator of late mitotic events. Research over the last decade has revealed many of its functions and today we know that this protein phosphatase orchestrates several aspects of chromosome segregation and is the key trigger of exit from mitosis. Elucidation of the mechanisms controlling Cdc14 activity through nucleolar sequestration now serves as a paradigm for how regulation of the subcellular localization of proteins regulates protein function. Here I review these findings focusing on how discoveries in my laboratory helped elucidate the function and regulation of Cdc14.
Figures



References
-
- Bloom J, Cross FR. Multiple levels of cyclin specificity in cell-cycle control. Nat Rev Mol Cell Bio. 2007;8(2):149–160. Review. - PubMed
-
- Murray AW, Solomon MJ, Kirschner MW. The role of cyclin synthesis and degradation in the control of maturation promoting factor activity. Nature. 1989;339(6222):280–286. - PubMed
-
- Irniger S, Piatti S, Michaelis C, Nasmyth K. Genes involved in sister chromatid separation are needed for B-type cyclin proteolysis in budding yeast. Cell. 1995;81(2):269–278. Erratum in: Cell 93(3), 487. - PubMed
-
- King RW, Peters JM, Tugendreich S, Rolfe M, Hieter P, Kirschner MW. A 20S complex containing CDC27 and CDC16 catalyzes the mitosis-specific conjugation of ubiquitin to cyclin B. Cell. 1995;81(2):279–288. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

