Reprogramming and differentiation in mammals: motifs and mechanisms
- PMID: 19022741
- PMCID: PMC2735112
- DOI: 10.1101/sqb.2008.73.016
Reprogramming and differentiation in mammals: motifs and mechanisms
Abstract
The natural reprogramming of the mammalian egg and sperm genomes is an efficient process that takes place in less than 24 hours and gives rise to a totipotent zygote. Transfer of somatic nuclei to mammalian oocytes also leads to their reprogramming and formation of totipotent embryos, albeit very inefficiently and requiring an activation step. Reprogramming of differentiated cells to induced pluripotent stem (iPS) cells takes place during a period of time substantially longer than reprogramming of the egg and sperm nuclei and is significantly less efficient. The stochastic expression of endogenous proteins during this process would imply that controlled expression of specific proteins is crucial for reprogramming to take place. The fact that OCT4, NANOG, and SOX2 form the core components of the pluripotency circuitry would imply that control at the transcriptional level is important for reprogramming to iPS cells. In contradistinction, the much more efficient reprogramming of the mammalian egg and sperm genomes implies that other levels of control are necessary, such as chromatin remodeling, translational regulation, and efficient degradation of no longer needed proteins and RNAs.
Figures

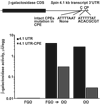
 Graph of reporter construct containing the intact CPE sequence;
Graph of reporter construct containing the intact CPE sequence;  Graph of reporter construct containing the mutated CPE sequence.
Graph of reporter construct containing the mutated CPE sequence.
References
-
- Adewumi O, Aflatoonian B, Ahrlund-Richter L, Amit M, Andrews PW, Beighton G, Bello PA, Benvenisty N, Berry LS, Bevan S, Blum B, Brooking J, Chen KG, Choo AB, Churchill GA, Corbel M, Damjanov I, Draper JS, Dvorak P, Emanuelsson K, Fleck RA, Ford A, Gertow K, Gertsenstein M, Gokhale PJ, Hamilton RS, Hampl A, Healy LE, Hovatta O, Hyllner J, Imreh MP, Itskovitz-Eldor J, Jackson J, Johnson JL, Jones M, Kee K, King BL, Knowles BB, Lako M, Lebrin F, Mallon BS, Manning D, Mayshar Y, McKay RD, Michalska AE, Mikkola M, Mileikovsky M, Minger SL, Moore HD, Mummery CL, Nagy A, Nakatsuji N, O'Brien CM, Oh SK, Olsson C, Otonkoski T, Park KY, Passier R, Patel H, Patel M, Pedersen R, Pera MF, Piekarczyk MS, Pera RA, Reubinoff BE, Robins AJ, Rossant J, Rugg-Gunn P, Schulz TC, Semb H, Sherrer ES, Siemen H, Stacey GN, Stojkovic M, Suemori H, Szatkiewicz J, Turetsky T, Tuuri T, van den Brink S, Vintersten K, Vuoristo S, Ward D, Weaver TA, Young LA, Zhang W. Characterization of human embryonic stem cell lines by the International Stem Cell Initiative. Nat Biotechnol. 2007;25:803. - PubMed
-
- Belloc E, Mendez R. A deadenylation negative feedback mechanism governs meiotic metaphase arrest. Nature. 2008;452:1017. - PubMed
-
- Bevilacqua A, Kinnunen LH, Mangia F. Genetic manipulation of mammalian dictyate oocytes: factors affecting transient expression of microinjected DNA templates. Mol Reprod Dev. 1992;33:124. - PubMed
-
- Bushati N, Stark A, Brennecke J, Cohen SM. Temporal reciprocity of miRNAs and their targets during the maternal-to-zygotic transition in Drosophila. Curr Biol. 2008;18:501. - PubMed
-
- Campbell KH, McWhir J, Ritchie WA, Wilmut I. Sheep cloned by nuclear transfer from a cultured cell line. Nature. 1996;380:64. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
