Pharmacological characterization of pannexin-1 currents expressed in mammalian cells
- PMID: 19023039
- PMCID: PMC2682283
- DOI: 10.1124/jpet.108.146365
Pharmacological characterization of pannexin-1 currents expressed in mammalian cells
Abstract
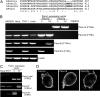
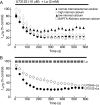
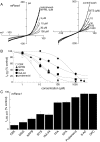
Pannexin (Panx) 1 is a widely expressed protein that shares structural, but not amino acid, homology with gap junction proteins, the connexins. Panx1 does not form gap junctions in mammalian cells, but it may function as a plasma membrane hemichannel. Little is known of the pharmacological properties of panx1 expression in mammalian cells. Here, we identify three variants in the human PANX1 gene. We expressed these variants and mouse Panx1 in mammalian cells and compared Panx1-induced currents. All human Panx1 variants and the mouse Panx1 showed identical protein expression levels, localization patterns, and functional properties, although the frequency of functional expression was species-dependent. Panx1 currents were independent of changes in extracellular or intracellular calcium or phospholipase C transduction. We found compounds that inhibited Panx1 currents with a rank order of potency: carbenoxolone > disodium 4,4'-diisothiocyanatostilbene-2,2'-disulfonate (DIDS) approximately disodium 4-acetamido-4'-isothiocyanato-stilben-2,2'-disulfonate approximately 5-nitro-2-(3-phenylpropylamino)benzoic acid > indanyloxyacetic acid 94 >> probenecid >> flufenamic acid = niflumic acid. Triphosphate nucleotides (ATP, GTP, and UTP) rapidly and reversibly inhibited Panx1 currents via mechanism(s) independent of purine receptors. When Panx1 was coexpressed with purinergic P2X(7) receptor (P2X(7)R), DIDS was found to act as a P2X(7)R antagonist to inhibit ATP-evoked currents, but none of the other compounds inhibited P2X(7)R currents. This is the first detailed pharmacological characterization of Panx1-mediated currents in mammalian cells and sheds new, although contradictory, light on the hypothesis that Panx1 acts as a hemichannel to allow passage of large molecules in response to P2X(7)R activation.
Figures




References
-
- Baranova A, Ivanov D, Petrash N, Pestova A, Skoblov M, Kelmanson I, Shagin D, Nazarenko S, Geraymovych E, Litvin O, et al. (2004) The mammalian pannexin family is homologous to the invertebrate pannexin gap junction proteins. Genomics 83 706-716. - PubMed
-
- Barbe MT, Monyer H, and Bruzzone R (2006) Cell-cell communication beyond connexins: the pannexin channels. Physiology 21 103-114. - PubMed
-
- Bruzzone R, Barbe MT, Jakob NJ, and Monyer H (2005) Pharmacological properties of homomeric and heteromeric pannexin hemichannels expressed in Xenopus oocytes. J Neurochem 92 1033-1043. - PubMed
-
- Gelband CH, Greco PG, and Martens JR (1996) Voltage-dependent chloride channels: invertebrates to man. J Exp Zool 275 277-282. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

