Flexible dosing of tincture of opium in the management of opioid withdrawal: pharmacokinetics and pharmacodynamics
- PMID: 19032172
- PMCID: PMC2661979
- DOI: 10.1111/j.1365-2125.2008.03277.x
Flexible dosing of tincture of opium in the management of opioid withdrawal: pharmacokinetics and pharmacodynamics
Abstract
Aims: The aim was to evaluate the clinical effectiveness, pharmacodynamics and pharmacokinetics of a range of Tincture of Opium (TOP) doses in the management of opioid withdrawal.
Methods: Forty-five opium-dependent Thai subjects were allocated to three dosing groups (6.66, 13.3 and 20 mg morphine equivalents, twice daily) depending on their self-reported prior opium use. On day 5 of dosing subjects underwent an interdosing interval study where blood, withdrawal scores, heart rate and blood pressure (BP) were collected at 0, 1, 3 and 8 h. Plasma morphine concentrations were quantified by high-performance liquid chromatography, and plasma morphine-3-glucuronide (M3G) and morphine-6-glucuronide (M6G) concentrations by LCMS.
Results: Thirty-two subjects completed the study. Withdrawal scores were low for all subjects (range 9-23% of maximum response). There were dose-dependent changes in both systolic and diastolic BP (P = 0.021 and P = 0.01, respectively), but these were not considered clinically significant. There were no effects of dose on respiratory rate. Plasma morphine concentrations changed significantly across the interdosing interval (P = 0.0001), rising to a maximum at 1 h after dosing. Plasma morphine concentrations also differed according to dose (P < 0.05). The mean ratios of the morphine glucuronides were found to be: M3G/M6G = 7.7, M3G/morphine = 35.6 and M6G/morphine = 4.9, values comparable to those previously reported.
Conclusion: The management of opioid withdrawal can be achieved, with minimal adverse effects, by using flexible dosing of TOP.
Figures
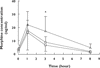
 ); Dose 2 (
); Dose 2 ( ); Dose 3 (
); Dose 3 ( )
)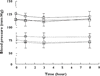
 ); SBP (Dose 2) (
); SBP (Dose 2) ( ); SBP (Dose 3) (
); SBP (Dose 3) ( ); DBP (Dose 1) (
); DBP (Dose 1) ( ); DBP (Dose 2) (
); DBP (Dose 2) ( ); DBP (Dose 3) (
); DBP (Dose 3) ( )
)
 ); Dose 2 (
); Dose 2 ( ); Dose 3 (
); Dose 3 ( )
)
 ); Dose 2 (
); Dose 2 ( ); Dose 3 (
); Dose 3 ( ) (B) Log withdrawal scores adjusted for prior daily opium use and plasma morphine concentration at each time point for all subjects. *P < 0.05; time point 1 h vs. 3 h
) (B) Log withdrawal scores adjusted for prior daily opium use and plasma morphine concentration at each time point for all subjects. *P < 0.05; time point 1 h vs. 3 hSimilar articles
-
Comparison of tincture of opium and methadone to control opioid withdrawal in a Thai treatment centre.Br J Clin Pharmacol. 2004 Nov;58(5):536-41. doi: 10.1111/j.1365-2125.2004.02209.x. Br J Clin Pharmacol. 2004. PMID: 15521902 Free PMC article. Clinical Trial.
-
High levels of morphine-6-glucuronide in street heroin addicts.Psychopharmacology (Berl). 2003 Nov;170(2):200-4. doi: 10.1007/s00213-003-1531-x. Epub 2003 May 28. Psychopharmacology (Berl). 2003. PMID: 12774191
-
Pharmacokinetic modelling of morphine, morphine-3-glucuronide and morphine-6-glucuronide in plasma and cerebrospinal fluid of neurosurgical patients after short-term infusion of morphine.Br J Clin Pharmacol. 2002 Dec;54(6):592-603. doi: 10.1046/j.1365-2125.2002.t01-1-01689.x. Br J Clin Pharmacol. 2002. PMID: 12492606
-
Morphine metabolites.Acta Anaesthesiol Scand. 1997 Jan;41(1 Pt 2):116-22. doi: 10.1111/j.1399-6576.1997.tb04625.x. Acta Anaesthesiol Scand. 1997. PMID: 9061094 Review.
-
Systematic review of factors affecting the ratios of morphine and its major metabolites.Pain. 1998 Jan;74(1):43-53. doi: 10.1016/S0304-3959(97)00142-5. Pain. 1998. PMID: 9514559
Cited by
-
Maintenance therapy with opium tincture for injecting drug users; implications for prevention from viral infections.Hepat Mon. 2013 Apr 16;13(4):e8334. doi: 10.5812/hepatmon.8334. Print 2013 Apr. Hepat Mon. 2013. PMID: 23805161 Free PMC article. No abstract available.
-
Comparing the Efficacy of Methadone and Tincture of Opium in Controlling Agitation Caused by Withdrawal Syndrome in Opium-Addicted Patients in the Intensive Care Unit: A Randomized Trial Study.Addict Health. 2020 Apr;12(2):69-76. doi: 10.22122/ahj.v12i2.259. Addict Health. 2020. PMID: 32782729 Free PMC article.
-
Methadone inhibits CYP2D6 and UGT2B7/2B4 in vivo: a study using codeine in methadone- and buprenorphine-maintained subjects.Br J Clin Pharmacol. 2012 May;73(5):786-94. doi: 10.1111/j.1365-2125.2011.04145.x. Br J Clin Pharmacol. 2012. PMID: 22092298 Free PMC article.
-
Does ingestion of tincture of opium notably raise blood alcohol concentration?Addict Health. 2014 Summer-Autumn;6(3-4):100-4. Addict Health. 2014. PMID: 25984276 Free PMC article.
-
Opium-associated QT Interval Prolongation: A Cross-sectional Comparative Study.Indian J Crit Care Med. 2021 Jan;25(1):43-47. doi: 10.5005/jp-journals-10071-23596. Indian J Crit Care Med. 2021. PMID: 33603300 Free PMC article.
References
-
- Holmstrand J, Anggård E, Gunne LM. Methadone maintenance: plasma levels and therapeutic outcome. Clin Pharmacol Ther. 1978;23:175–80. - PubMed
-
- Schall U, Pries E, Katta T, Kloppel A, Gastpar M. Pharmacokinetic and pharmacodynamic interactions in an outpatient maintenance therapy of intravenous heroin users with levomethadone. Addict Biol. 1996;1:105–13. - PubMed
-
- Ward J, Hall W, Mattick RP. Role of maintenance treatment in opioid dependence. Lancet. 1999;353:221–6. - PubMed
-
- Bell J, Mattick R, Hay A, Chan J, Hall W. Methadone maintenance and drug-related crime. J Subst Abuse. 1997;9:15–25. - PubMed
-
- Grøndbladh L, Ohlund LS, Gunne LM. Mortality in heroin addiction: impact of methadone treatment. Acta Psychiatr Scand. 1990;82:223–7. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

