Atheroprotective effects of methotrexate on reverse cholesterol transport proteins and foam cell transformation in human THP-1 monocyte/macrophages
- PMID: 19035488
- PMCID: PMC2599810
- DOI: 10.1002/art.24040
Atheroprotective effects of methotrexate on reverse cholesterol transport proteins and foam cell transformation in human THP-1 monocyte/macrophages
Abstract
Objective: To determine whether methotrexate (MTX) can overcome the atherogenic effects of cyclooxygenase 2 (COX-2) inhibitors and interferon-gamma (IFNgamma), both of which suppress cholesterol efflux protein and promote foam cell transformation in human THP-1 monocyte/macrophages.
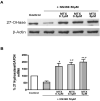
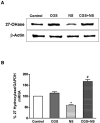
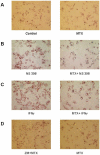
Methods: Message and protein levels of the reverse cholesterol transport proteins cholesterol 27-hydroxylase and ATP-binding cassette transporter A1 (ABCA1) in THP-1 cells were evaluated by real-time polymerase chain reaction and immunoblot, respectively. Expression was evaluated in cells incubated in the presence or absence of the COX-2 inhibitor NS398 or IFNgamma, with and without MTX. Foam cell transformation of lipid-laden THP-1 macrophages was detected with oil red O staining and light microscopy.
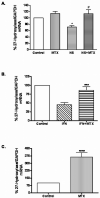
Results: MTX increased 27-hydroxylase message and completely blocked NS398-induced down-regulation of 27-hydroxylase (mean +/- SEM 112.8 +/- 13.1% for NS398 plus MTX versus 71.1 +/- 4.3% for NS398 alone; P < 0.01). MTX also negated COX-2 inhibitor-mediated down-regulation of ABCA1. The ability of MTX to reverse inhibitory effects on 27-hydroxylase and ABCA1 was blocked by the adenosine A2A receptor-specific antagonist ZM241385. MTX also prevented NS398 and IFNgamma from increasing transformation of lipid-laden THP-1 macrophages into foam cells.
Conclusion: This study provides evidence supporting the notion of an atheroprotective effect of MTX. Through adenosine A2A receptor activation, MTX promotes reverse cholesterol transport and limits foam cell formation in THP-1 macrophages. This is the first reported evidence that any commonly used medication can increase expression of antiatherogenic reverse cholesterol transport proteins and can counteract the effects of COX-2 inhibition. Our results suggest that one mechanism by which MTX protects against cardiovascular disease in rheumatoid arthritis patients is through facilitation of cholesterol outflow from cells of the artery wall.
Figures


Similar articles
-
Effect of cyclooxygenase inhibition on cholesterol efflux proteins and atheromatous foam cell transformation in THP-1 human macrophages: a possible mechanism for increased cardiovascular risk.Arthritis Res Ther. 2007;9(1):R4. doi: 10.1186/ar2109. Arthritis Res Ther. 2007. PMID: 17244362 Free PMC article.
-
Adenosine A2A receptor occupancy stimulates expression of proteins involved in reverse cholesterol transport and inhibits foam cell formation in macrophages.J Leukoc Biol. 2004 Sep;76(3):727-34. doi: 10.1189/jlb.0204107. Epub 2004 Jun 14. J Leukoc Biol. 2004. PMID: 15197231
-
Blood lipid profiles and peripheral blood mononuclear cell cholesterol metabolism gene expression in patients with and without methotrexate treatment.BMC Med. 2011 Jan 13;9:4. doi: 10.1186/1741-7015-9-4. BMC Med. 2011. PMID: 21232092 Free PMC article.
-
Foam cells in atherosclerosis.Clin Chim Acta. 2013 Sep 23;424:245-52. doi: 10.1016/j.cca.2013.06.006. Epub 2013 Jun 16. Clin Chim Acta. 2013. PMID: 23782937 Review.
-
Regulation of foam cells by adenosine.Arterioscler Thromb Vasc Biol. 2012 Apr;32(4):879-86. doi: 10.1161/ATVBAHA.111.226878. Arterioscler Thromb Vasc Biol. 2012. PMID: 22423040 Free PMC article. Review.
Cited by
-
Macrophage Dysfunction in Autoimmune Rheumatic Diseases and Atherosclerosis.Int J Mol Sci. 2022 Apr 19;23(9):4513. doi: 10.3390/ijms23094513. Int J Mol Sci. 2022. PMID: 35562903 Free PMC article. Review.
-
Double trouble: psoriasis and cardiometabolic disorders.Cardiovasc J Afr. 2018 May/Jun 23;29(3):189-194. doi: 10.5830/CVJA-2017-055. Epub 2017 Dec 13. Cardiovasc J Afr. 2018. PMID: 29293257 Free PMC article. Review.
-
A new weapon against an old target.Arthritis Res Ther. 2011 Aug 12;13(4):122. doi: 10.1186/ar3392. Arthritis Res Ther. 2011. PMID: 21861856 Free PMC article.
-
Cardiovascular Death Risk in Primary Central Nervous System Lymphoma Patients Treated With Chemotherapy: A Registry-Based Cohort Study.Front Oncol. 2021 May 11;11:641955. doi: 10.3389/fonc.2021.641955. eCollection 2021. Front Oncol. 2021. PMID: 34046345 Free PMC article.
-
Prevention of stroke in rheumatoid arthritis.Curr Treat Options Neurol. 2015 Jul;17(7):356. doi: 10.1007/s11940-015-0356-3. Curr Treat Options Neurol. 2015. PMID: 25981388
References
-
- Ward JR. Historical perspective on the use of methotrexate for the treatment of rheumatoid arthritis. J Rheumatol. 1985;12(suppl 12):3–6. - PubMed
-
- Kremer JM. Historical overview of the treatment of rheumatoid arthritis with an emphasis on methotrexate. J Rheumatol. 1996;44:34–7. - PubMed
-
- Alarcon GS. Methotrexate use in rheumatoid arthritis. A clinician's perspective. Immunopharmacology. 2000;47:259–71. - PubMed
-
- Moss RB. Alternative pharmacotherapies for steroid-dependent asthma. Chest. 1995;107:817–25. - PubMed
-
- Wong JM, Esdaile JM. Methotrexate in systemic lupus erythematosus. Lupus. 2005;14:101–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

