Loss of LR11/SORLA enhances early pathology in a mouse model of amyloidosis: evidence for a proximal role in Alzheimer's disease
- PMID: 19036982
- PMCID: PMC2669320
- DOI: 10.1523/JNEUROSCI.4582-08.2008
Loss of LR11/SORLA enhances early pathology in a mouse model of amyloidosis: evidence for a proximal role in Alzheimer's disease
Abstract
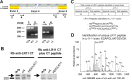
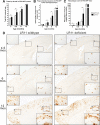
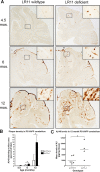
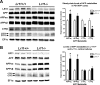
Alzheimer's disease (AD) is the most prevalent form of dementia, resulting in progressive neuronal death and debilitating damage to brain loci that mediate memory and higher cognitive function. While pathogenic genetic mutations have been implicated in approximately 2% of AD cases, the proximal events that underlie the common, sporadic form of the disease are incompletely understood. Converging lines of evidence from human neuropathology, basic biology, and genetics have implicated loss of the multifunctional receptor LR11 (also known as SORLA and SORL1) in AD pathogenesis. Cell-based studies suggest that LR11 reduces the formation of beta-amyloid (Abeta), the molecule believed to be a primary toxic species in AD. Recently, mutant mice deficient in LR11 were shown to upregulate murine Abeta in mouse brain. In the current study, LR11-deficient mice were crossed with transgenic mice expressing autosomal-dominant human AD genes, presenilin-1 (PS1DeltaE9) and amyloid precursor protein (APPswe). Here, we show that LR11 deficiency in this AD mouse model significantly increases Abeta levels and exacerbates early amyloid pathology in brain, causing a forward shift in disease onset that is LR11 gene dose-dependent. Loss of LR11 increases the processing of the APP holo-molecule into alpha-, beta-, and gamma-secretase derived metabolites. We propose that LR11 regulates APP processing and Abeta accumulation in vivo and is of proximal importance to the cascade of pathological amyloidosis. The results of the current study support the hypothesis that control of LR11 expression may exert critical effects on Alzheimer's disease susceptibility in humans.
Figures


References
-
- Andersen OM, Reiche J, Schmidt V, Gotthardt M, Spoelgen R, Behlke J, von Arnim CA, Breiderhoff T, Jansen P, Wu X, Bales KR, Cappai R, Masters CL, Gliemann J, Mufson EJ, Hyman BT, Paul SM, Nykjaer A, Willnow TE. Neuronal sorting protein-related receptor sorLA/LR11 regulates processing of the amyloid precursor protein. Proc Natl Acad Sci U S A. 2005;102:13461–13466. - PMC - PubMed
-
- Andersen OM, Schmidt V, Spoelgen R, Gliemann J, Behlke J, Galatis D, McKinstry WJ, Parker MW, Masters CL, Hyman BT, Cappai R, Willnow TE. Molecular dissection of the interaction between amyloid precursor protein and its neuronal trafficking receptor SorLA/LR11. Biochemistry. 2006;45:2618–2628. - PubMed
-
- Bettens K, Brouwers N, Engelborghs S, De Deyn PP, Van Broeckhoven C, Sleegers K. SORL1 is genetically associated with increased risk for late-onset Alzheimer disease in the Belgian population. Hum Mutat. 2008;29:769–770. - PubMed
-
- Böhm C, Seibel NM, Henkel B, Steiner H, Haass C, Hampe W. SorLA signaling by regulated intramembrane proteolysis. J Biol Chem. 2006;281:14547–14553. - PubMed
-
- Borchelt DR, Thinakaran G, Eckman CB, Lee MK, Davenport F, Ratovitsky T, Prada CM, Kim G, Seekins S, Yager D, Slunt HH, Wang R, Seeger M, Levey AI, Gandy SE, Copeland NG, Jenkins NA, Price DL, Younkin SG, Sisodia SS. Familial Alzheimer's disease-linked presenilin 1 variants elevate Abeta1–42/1–40 ratio in vitro and in vivo. Neuron. 1996;17:1005–1013. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
