Inhibiting the function of ABCB1 and ABCG2 by the EGFR tyrosine kinase inhibitor AG1478
- PMID: 19059384
- PMCID: PMC2843910
- DOI: 10.1016/j.bcp.2008.11.007
Inhibiting the function of ABCB1 and ABCG2 by the EGFR tyrosine kinase inhibitor AG1478
Abstract
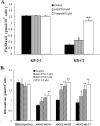
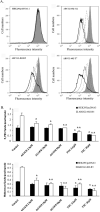
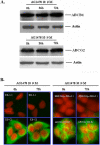
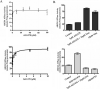
The tyrphostin 4-(3-chloroanilino)-6,7-dimethoxyquinazoline (AG1478) is a potent and specific EGFR tyrosine kinase inhibitor (TKI); its promising pre-clinical results have led to clinical trials. Overexpression of ATP-binding cassette (ABC) transporters such as ABCB1, ABCC1 and ABCG2 is one of the main causes of multidrug resistance (MDR) and usually results in the failure of cancer chemotherapy. However, the interaction of AG1478 with these ABC transporters is still unclear. In the present study, we have investigated this interaction and found that AG1478 has differential effects on these transporters. In ABCB1-overexpressing cells, non-toxic doses of AG1478 were found to partially inhibit resistance to ABCB1 substrate anticancer drugs as well as increase intracellular accumulation of [3H]-paclitaxel. Similarly, in ABCG2-overexpressing cells, AG1478 significantly reversed resistance to ABCG2 substrate anticancer drugs and increased intracellular accumulation of [3H]-mitoxantrone as well as fluorescent compound BODIPY-prazosin. AG1478 also profoundly inhibited the transport of [3H]-E(2)17betaG and [3H]-methotrexate by ABCG2. We also found that AG1478 slightly stimulated ABCB1 ATPase activity and significantly stimulated ABCG2 ATPase activity. Interestingly, AG1478 did not inhibit the photolabeling of ABCB1 or ABCG2 with [125I]-iodoarylazidoprazosin. Additionally, AG1478 did not alter the sensitivity of parental, ABCB1- or ABCG2-overexpressing cells to non-ABCB1 and non-ABCG2 substrate drug and had no effect on the function of ABCC1. Overall, we conclude that AG1478 is able to inhibit the function of ABCB1 and ABCG2, with a more pronounced effect on ABCG2. Our findings provide valuable contributions to the development of safer and more effective EGFR TKIs for use as anticancer agents in the clinic.
Figures

Similar articles
-
Lapatinib (Tykerb, GW572016) reverses multidrug resistance in cancer cells by inhibiting the activity of ATP-binding cassette subfamily B member 1 and G member 2.Cancer Res. 2008 Oct 1;68(19):7905-14. doi: 10.1158/0008-5472.CAN-08-0499. Cancer Res. 2008. PMID: 18829547 Free PMC article.
-
Erlotinib (Tarceva, OSI-774) antagonizes ATP-binding cassette subfamily B member 1 and ATP-binding cassette subfamily G member 2-mediated drug resistance.Cancer Res. 2007 Nov 15;67(22):11012-20. doi: 10.1158/0008-5472.CAN-07-2686. Cancer Res. 2007. PMID: 18006847
-
Sildenafil reverses ABCB1- and ABCG2-mediated chemotherapeutic drug resistance.Cancer Res. 2011 Apr 15;71(8):3029-41. doi: 10.1158/0008-5472.CAN-10-3820. Epub 2011 Mar 14. Cancer Res. 2011. PMID: 21402712 Free PMC article.
-
Variation and evolution of the ABC transporter genes ABCB1, ABCC1, ABCG2, ABCG5 and ABCG8: implication for pharmacogenetics and disease.Drug Metabol Drug Interact. 2011;26(4):169-79. doi: 10.1515/DMDI.2011.027. Epub 2011 Nov 18. Drug Metabol Drug Interact. 2011. PMID: 22098604 Free PMC article. Review.
-
In silico prediction of substrate properties for ABC-multidrug transporters.Expert Opin Drug Metab Toxicol. 2008 Sep;4(9):1167-80. doi: 10.1517/17425255.4.9.1167. Expert Opin Drug Metab Toxicol. 2008. PMID: 18721111 Review.
Cited by
-
Carvacrol Alleviates Prostate Cancer Cell Proliferation, Migration, and Invasion through Regulation of PI3K/Akt and MAPK Signaling Pathways.Oxid Med Cell Longev. 2016;2016:1469693. doi: 10.1155/2016/1469693. Epub 2016 Oct 10. Oxid Med Cell Longev. 2016. PMID: 27803760 Free PMC article.
-
GSK2606414 Sensitizes ABCG2-Overexpressing Multidrug-Resistant Colorectal Cancer Cells to Chemotherapeutic Drugs.Biomedicines. 2023 Nov 20;11(11):3103. doi: 10.3390/biomedicines11113103. Biomedicines. 2023. PMID: 38002103 Free PMC article.
-
Role of HDAC3-miRNA-CAGE Network in Anti-Cancer Drug-Resistance.Int J Mol Sci. 2018 Dec 23;20(1):51. doi: 10.3390/ijms20010051. Int J Mol Sci. 2018. PMID: 30583572 Free PMC article. Review.
-
Polycaprolactone Electrospun Scaffolds Produce an Enrichment of Lung Cancer Stem Cells in Sensitive and Resistant EGFRm Lung Adenocarcinoma.Cancers (Basel). 2021 Oct 22;13(21):5320. doi: 10.3390/cancers13215320. Cancers (Basel). 2021. PMID: 34771484 Free PMC article.
-
2-Trifluoromethyl-2-Hydroxypropionamide Derivatives as Novel Reversal Agents of ABCG2 (BCRP)-Mediated Multidrug Resistance: Synthesis and Biological Evaluations.J Cell Biochem. 2017 Aug;118(8):2420-2429. doi: 10.1002/jcb.25908. Epub 2017 Apr 25. J Cell Biochem. 2017. PMID: 28120346 Free PMC article.
References
-
- Levitzki A, Gazit A. Tyrosine kinase inhibition: an approach to drug development. Science. 1995;267:1782–8. - PubMed
-
- Arteaga CL, Ramsey TT, Shawver LK, Guyer CA. Unliganded epidermal growth factor receptor dimerization induced by direct interaction of quinazolines with the ATP binding site. J Biol Chem. 1997;272:23247–54. - PubMed
-
- Gazit A, Chen J, App H, McMahon G, Hirth P, Chen I, et al. Tyrphostins IV--highly potent inhibitors of EGF receptor kinase. Structure-activity relationship study of 4-anilidoquinazolines. Bioorg Med Chem. 1996;4:1203–7. - PubMed
-
- Bridges AJ. The rationale and strategy used to develop a series of highly potent, irreversible, inhibitors of the epidermal growth factor receptor family of tyrosine kinases. Curr Med Chem. 1999;6:825–43. - PubMed
-
- Partik G, Hochegger K, Schorkhuber M, Marian B. Inhibition of epidermal-growth-factor-receptor-dependent signalling by tyrphostins A25 and AG1478 blocks growth and induces apoptosis in colorectal tumor cells in vitro. J Cancer Res Clin Oncol. 1999;125:379–88. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

