NAD(P)H quinone oxidoreductase 1 is essential for ozone-induced oxidative stress in mice and humans
- PMID: 19059883
- PMCID: PMC2701957
- DOI: 10.1165/rcmb.2008-0381OC
NAD(P)H quinone oxidoreductase 1 is essential for ozone-induced oxidative stress in mice and humans
Erratum in
-
Erratum: NAD(P)H Quinone Oxidoreductase 1 Is Essential for Ozone-Induced Oxidative Stress in Mice and Humans.Am J Respir Cell Mol Biol. 2016 Dec;55(6):909. doi: 10.1165/rcmb.556erratum. Am J Respir Cell Mol Biol. 2016. PMID: 27906558 Free PMC article. No abstract available.
Abstract
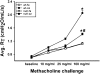
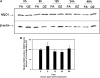
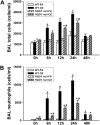
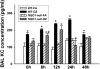
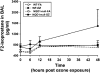
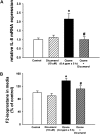
One host susceptibility factor for ozone identified in epidemiologic studies is NAD(P)H quinone oxidoreductase 1 (NQO1). We hypothesized that after ozone exposure, NQO1 is required to increase 8-isoprostane (also known as F(2)-isoprostane) production, a recognized marker of ozone-induced oxidative stress, and to enhance airway inflammation and hyperresponsiveness. In this report, we demonstrate that in contrast to wild-type mice, NQO1-null mice are resistant to ozone and have blunted responses, including decreased production of F(2)-isoprostane and keratinocyte chemokine, decreased airway inflammation, and diminished airway hyperresponsiveness. Importantly, these results in mice correlate with in vitro findings in humans. In primary human airway epithelial cells, inhibition of NQO1 by dicumarol blocks ozone-induced F(2)-isoprostane production and IL-8 gene expression. Together, these results demonstrate that NQO1 modulates cellular redox status and influences the biologic and physiologic effects of ozone.
Figures
Comment in
-
Findings of Research Misconduct.Fed Regist. 2019 Nov 7;84(216):60097-60098. Fed Regist. 2019. PMID: 37547121 Free PMC article. No abstract available.
References
-
- American Thoracic Society. Health effects of outdoor air pollution. Part 2. Committee of the Environmental and Occupational Health Assembly of the American Thoracic Society. Am J Respir Crit Care Med 1996;153:477–498. - PubMed
-
- Peden DB. The epidemiology and genetics of asthma risk associated with air pollution. J Allergy Clin Immunol 2005;115:213–219. (quiz 220). - PubMed
-
- Foster W. Effects of oxidants. In: Swift D, Foster W, editors. Air pollutants and the respiratory tract. New York: Marcel Dekker; 1999. pp. 147–179.
-
- Trasande L, Thurston GD. The role of air pollution in asthma and other pediatric morbidities. J Allergy Clin Immunol 2005;115:689–699. - PubMed
-
- Gent JF, Triche EW, Holford TR, Belanger K, Bracken MB, Beckett WS, Leaderer BP. Association of low-level ozone and fine particles with respiratory symptoms in children with asthma. JAMA 2003;290:1859–1867. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

